Stem Cells Finally Deliver, But Not on Their Original Promise
Stem cell therapies remain few and far between, but the versatile tissues are transforming how we understand disease.

To scientists, stem cells represent the potential of the human body to heal itself. The cells are our body’s wide-eyed kindergarteners—they have the potential to do pretty much anything, from helping us obtain oxygen, digest food, or pump our blood. That flexibility has given scientists hope that they can coax stem cells to differentiate into and replace those damaged by illness.
Almost immediately after scientists learned how to isolate stem cells from human embryos, the excitement was palpable. In the lab, they had already been coaxed into becoming heart muscle, bone marrow, and kidney cells. Entire companies were founded to translate therapies into clinical trials. Nearly 20 years on, though, only a handful of therapies using stem cells have been approved. Not quite the revolution we had envisioned back in 1998.
Only a handful of therapies using stem cells have been approved.
But stem cells have delivered on another promise, one that is already having a broad impact on medical science. In their investigations into the potential therapeutic functions of stem cells, scientists have discovered another way to help those suffering from neurodegenerative and other incurable diseases. With stem cells, researchers can study how these diseases begin and even test the efficacy of drugs on cells from the very people they’re intended to treat.
Getting to this point hasn’t been easy. Research into pluripotent stem cells, the most promising type, has faced a number of scientific and ethical hurdles. They were most readily found in developing embryos, but in 1995, Congress passed a bill that eliminated funding on embryonic stem cells. Since adult humans don’t have pluripotent stem cells, researchers were stuck.
That changed in 2006, when Japanese scientist Shinya Yamanaka developed a way to create stem cells from a skin biopsy. Yamanaka’s process to create induced pluripotent stem cells (iPS cells) won him and his colleague John Gurdon a Nobel Prize in 2012. After years of setbacks, the stem cell revolution was back on.
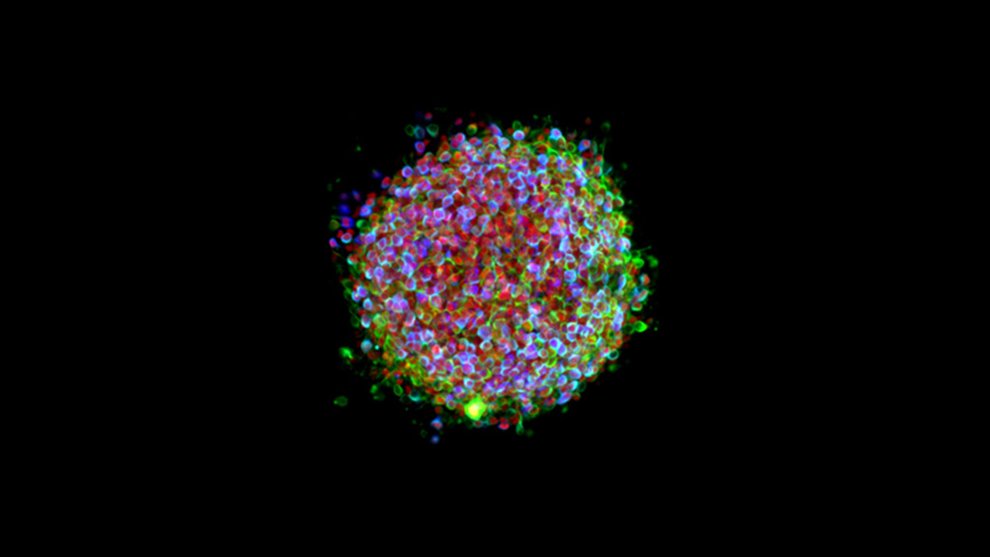
Biomedical scientists in fields from cancer to heart disease have turned to iPS cells in their research. But the technique has been especially popular among scientists studying neurodegenerative diseases like Alzheimer’s disease, Parkinson’s disease, and amyotrophic lateral sclerosis (ALS) for two main reasons: One, since symptoms of these diseases don’t develop until rather late in the disease process, scientists haven’t had much knowledge about the early stages. IPS cells changed that by allowing scientists to study the very early stages of the disorder. And two, they provide novel ways of testing new drugs and potentially even personalizing treatment options.
“It’s creating a sea change,” says Jeanne Loring, a stem cell biologist at the Scripps Research Institute in San Diego. “There will be tools available that have never been available before, and it will completely change drug development.”
Beyond Animal Models
Long before scientists knew that stem cells existed, they relied on animals to model diseases. Through careful breeding and, later, genetic engineering, researchers have developed rats, mice, fruit flies, roundworms, and other animals that display symptoms of the illness in question. Animal models remain useful, but they’re not perfect. While the biology of these animals often mimics humans’, they aren’t identical, and although some animals might share many of the overt symptoms of human illness, scientists can’t be sure that they experience the disease in the same way humans do.
“Mouse models are useful research tools, but they rarely capture the disease process,” says Rick Livesey , a biologist at the University of Cambridge in the U.K. Many neurodegenerative diseases, like Alzheimer’s, he says, are perfect examples of the shortcomings of animal models. “No other species of animal actually gets Alzheimer’s disease, so any animal model is a compromise.”
As a result, many drugs that seemed to be effective in animal models showed no benefit in humans. A study published in Alzheimer’s Research and Therapy in June 2014 estimated that 99.9% of Alzheimer’s clinical trials ended in failure, costing both money and lives. Scientists like Ole Isacson, a neuroscientist at Harvard University who studies Parkinson’s disease, were eager for a method that would let them investigate illnesses in a patient’s own cells, eliminating the need for expensive and imperfect animal models.
“No other species of animal actually gets Alzheimer’s disease, so any animal model is a compromise.”
Stem cells appeared to offer that potential, but when Congress banned federal funding in 1995 for research on embryos—and thus the development of new stem cell lines—scientists found their work had ground to a halt. As many researchers in the U.S. fretted over the future of stem cell research, scientists in Japan were developing a technique which would eliminate the need for embryonic stem cells. What’s more, it would allow researchers to create stem cells from the individuals who were suffering from the diseases they were studying.
Cells in the body are able to specialize by turning on some sets of genes and switching off others. Every cell has a complete copy of the DNA, it’s just packed away in deep storage where the cell can’t easily access it. Yamanaka, the Nobel laureate, knew that finding the key to this storage locker and unpacking it could potentially turn any specialized cell back into a pluripotent stem cell. He focused in on a group of 24 genes that were active only in embryonic stem cells. If he could get adult, specialized cells to translate these genes into proteins, then they should revert to stem cells. Yamanaka settled on fibroblast cells as the source of iPS cells since these are easily obtained with a skin biopsy.
Rather than trying to switch these genes back on, a difficult and time-consuming task, Yamanaka instead engineered a retrovirus to carry copies of these 24 genes to mouse fibroblast cells. Since many retroviruses insert their own genetic material into the genomes of the cells they infect, Yamanaka only had to deliver the virus once. All successive generations of cells inherited those 24 genes. Yamanaka first grew the fibroblasts in a dish, then infected them with his engineered retrovirus. Over repeated experiments, Yamanaka was able to narrow the suite of required genes from 24 down to just four.
The process was far from perfect—it took several weeks to create the stem cells, and only around 0.01%–0.1% of the fibroblasts were actually converted to stem cells. But after Yamanaka published his results in Cell in 2006, scientists quickly began perfecting the procedure and developing other techniques. To say they have been successful would be an understatement. “The technology is so good now that I have the undergraduates in my lab doing the reprogramming,” Loring says.
Accelerating Disease
When he heard of Yamanaka’s discovery, Isacson, the Harvard neuroscientist studying Parkinson’s disease, had been using fetal neurons to try to replace diseased and dying neurons. Isacson realized “very quickly” that iPS cells could yield new discoveries about Parkinson’s. At the time, scientists were trying to determine exactly when the disease process started. It wasn’t easy. A person has to lose around 70% of their dopamine neurons before the first sign of movement disorder appears and Parkinson’s can be diagnosed. By that point, it’s too late to reverse that damage, a problem that is found in many if not all neurodegenerative diseases. Isacson wanted to know what was causing the neurons to die.
Together with the National Institute of Neurological Disorders and Stroke consortium on iPS cells, Isacson obtained fibroblasts from patients with genetic mutations linked to Parkinson’s. Then, he reprogrammed these cells to become the specific type of neurons affected by Parkinson’s disease. “To our surprise, in the very strong hereditary forms of disease, we found that cells showed very strong signs of distress in the dish, even though they were newborn cells,” Isacson says.
These experiments, published in Science Translational Medicine in 2012, showed that the disease process in Parkinson’s started far earlier than scientists expected. The distressed, differentiated neurons Isacson saw under the microscope were still just a few weeks old. People generally didn’t start showing symptoms for Parkinson’s disease until middle age or beyond.
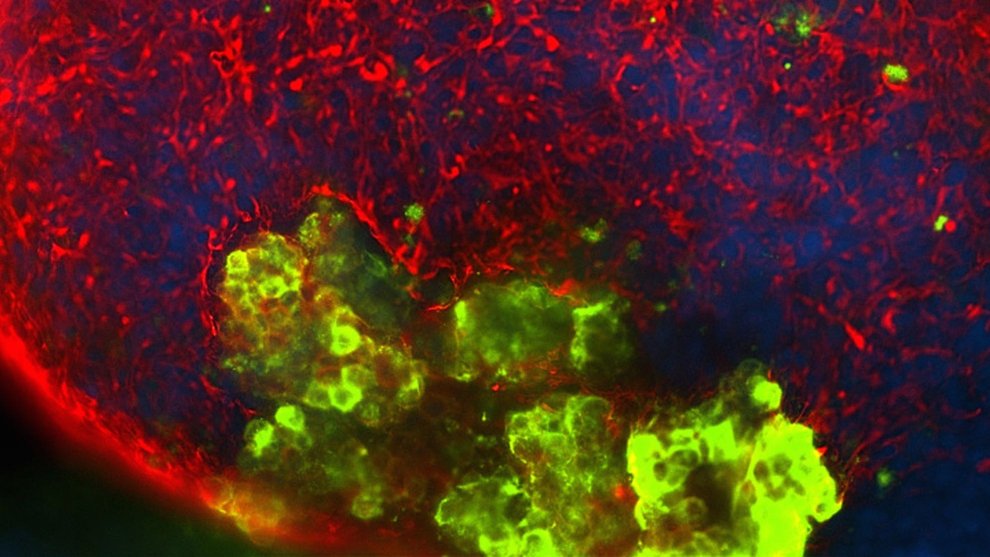
Isacson and his colleagues then tried to determine what was different between different cells with different mutations. The cells showed the most distress in their mitochondria, the parts of the cell that act as power plants by creating energy from oxygen and glucose. How that distress manifested, though, varied slightly depending on which mutation the patient carried. Neurons derived from an individual with a mutation in the LRRK2 gene consumed lower than expected amounts of oxygen, whereas the neurons derived from those carrying a mutation in PINK1 had much higher oxygen consumption. Neurons with these mutations were also more susceptible to a type of cellular damage known as oxidative stress.
After exposing both groups of cells to a variety of environmental toxins, such as oligomycin and valinomycin, both of which affect mitochondria, Isacson and colleagues attempted to rescue the cells by using several compounds that had been found effective in animal models. Both the LRRK2 and the PINK1 cells responded well to the antioxidant coenzyme Q10, but had very different responses to the immunosuppressant drug rapamycin. Whereas LRRK2 showed beneficial responses to rapamycin, the PINK1 cells did not.
To Isacson, the different responses were profoundly important. “Most drugs don’t become blockbusters because they don’t work for everyone. Trials start too late, and they don’t know the genetic background of the patient,” Isacson says. He believes that iPS cells will one day help researchers match specific treatments with specific genotypes. There may not be a single blockbuster that can treat Parkinson’s, but there may be several drugs that make meaningful differences in patients’ lives.
Cancer biologists have already begun culturing tumor cells and testing anti-cancer drugs before giving these medications to patients, and biologists studying neurodegenerative disease hope that iPS cells will one day allow them to do something similar for their patients. Scientists studying ALS have recently taken a step in that direction, using iPS cells to create motor neurons from fibroblasts of people carrying a mutation in the C9orf72 gene, the most common genetic cause of ALS. In a recent paper in Neuron , the scientists identified a small molecule which blocked the formation of toxic proteins caused by this mutation in cultured motor neurons.
Adding More Dimensions
It’s one thing to identify early disease in iPS cells, but these cells are generally obtained from people who have been diagnosed. At that point, it’s too late, in a way; drugs may be much less likely to work in later stages of the disease. To make many potential drugs more effective, the disease has to be diagnosed much, much earlier. Recent work by Harvard University stem cell biologist Rudolph Tanzi and colleagues may have taken a step in that direction, also using iPS cells.
Doo Yeon Kim, Tanzi’s co-author, had grown frustrated with iPS cell models of neurodegenerative disease. The cell cultures were liquid, and the cells could only grow in a thin, two-dimensional layer. The brain, however, was more gel-like, and existed in three dimensions. So Kim created a 3D gel matrix on which the researchers grew human neural stem cells that carried extra copies of two genes—one which codes for amyloid precursor protein and another for presenilin 1, both of which were previously discovered in Tanzi’s lab—which are linked to familial forms of Alzheimer’s disease.
After six weeks, the cells contained high levels of the harmful beta-amyloid protein as well as large numbers of toxic neurofibrillary tangles that damage and kill neurons. Both of these proteins had been found at high levels in the neurons of individuals who had died from Alzheimer’s disease, but researchers didn’t know for certain which protein built up first and which was more central to the disease process. Further experiments revealed that drugs preventing the formation of amyloid proteins also prevented the formation of neurofibrillary tangles, indicating that amyloid proteins likely formed first during Alzheimer’s disease.
“When you stop amyloid, you stop cell death,” Tanzi says. Amyloid begins to build up long before people show signs of altered cognition, and Tanzi believes that drugs which stop amyloid or prevent the buildup of neurofibrillary tangles could prevent Alzheimer’s before it starts.
The results were hailed in the media as a “major breakthrough,” although Larry Goldstein, a neuroscientist at the University of California, San Diego, takes a more nuanced perspective. “It’s a nice paper and an important step forward, but things got overblown. I don’t know that I would use the word ‘breakthrough’ because these, like all results, often have a very long history to them,” Goldstein says.
The scientists who spoke with NOVA Next about iPS cells noted that the field is moving forward at a remarkable clip, but they all talked at length about the issues that still remain. One of the largest revolves around differences between the age of the iPS cells and the age of the humans who develop these neurodegenerative diseases. Although scientists are working with neurons that are technically “mature,” they are nonetheless only weeks or months old—far from the several decades that the sufferers of neurodegenerative diseases have. Since aging remains the strongest risk factor for developing these diseases, neuroscientists worry that some disease pathology might be missed in such young cells. “Is it possible to study a disease that takes 70 years to develop in a person using cells that have grown for just a few months in a dish?” Livesey asks.
So far, the answer has been a tentative yes. Some scientists have begun to devise different strategies to accelerate the aging process in the lab so researchers don’t have to wait several decades before they develop their answers. Lorenz Studer, director of the Center for Stem Cell Biology at the Sloan-Kettering Institute, uses the protein that causes progeria, a disorder of extreme premature aging, to successfully age neurons derived from iPS cells from Parkinson’s disease patients.
“Is it possible to study a disease that takes 70 years to develop in a person using cells that have grown for just a few months in a dish?”
Robert Lanza, a stem cell biologist at Advanced Cell Technology, takes another approach, aging cells by taking small amounts of mature neurons and growing them up in a new dish. “Each time you do this, you are forcing the cells to divide,” Lanza says. “And cells can only divide so many times before they reach senescence and die.” This process, Lanza believes, will mimic aging. He has also been experimenting with stressing the cells to promote premature aging.
All of these techniques, Livesey believes, will allow scientists to study which aspects of the aging process—such as number of cell divisions and different types of environmental stressors—affect neurodegenerative diseases and how they do so. Adding to the complexity of the experimental system will improve the results that come out at the end. “You can only capture as much biology in iPS cells as you plug into it in the beginning,” Livesey says.
But as Isacson and Loring’s work, has shown, even very young cells can show hallmarks of neurodegenerative diseases. “If a disease has a genetic cause, if there’s an actual change in DNA, you should be able to find something in those iPS cells that is different,” Loring says.
For these experiments and others, scientists have been relying on iPS cells derived from individuals with hereditary or familial forms of neurodegenerative disease. These individuals, however, only represent about 5–15% of individuals with neurodegenerative disease; the vast majority of neurodegenerative diseases is sporadic and has no known genetic cause. Scientists believe that environmental factors may play a much larger role in the onset of these forms of neurodegenerative disease.
That heterogeneity means it’s not yet clear whether the iPS cells from individuals with hereditary forms of disease are a good model for what happens in sporadic disease. Although the resulting symptoms may be the same, different forms of disease may use the same biological pathways to end up in the same place. Isacson is in the process of identifying the range of genes and proteins that are altered in iPS cells that carry Parkinson’s disease mutations. He intends to determine whether any of these pathways are also disturbed in sporadically occurring Parkinson’s disease to pinpoint any similarities in both forms of disease.
Livesey’s lab just received a large grant to study people with an early onset, sporadic form of Alzheimer’s. “Although sporadic Alzheimer’s disease isn’t caused by a mutation in a single gene, the condition is still strongly heritable. The environment, obviously, has an important role, but so does genetics,” Livesey says.
Because the disease starts earlier in these individuals, researchers believe that it has a larger genetic link than other forms of sporadic Alzheimer’s disease, which will make it easier to identify any genetic or biological abnormalities. Livesey hopes that bridging sporadic and hereditary forms of Alzheimer’s disease will allow researchers to reach stronger conclusions using iPS cells.
Though it will be years before any new drugs come out of Livesey’s stem cell studies—or any other stem cell study for that matter—the technology has nonetheless allowed scientists to refine their understanding of these and other diseases. And, scientists believe, this is just the start. “There are an endless series of discoveries that can be made in the next few decades,” Isacson says.
Image credit: Ole Isacson, McLean Hospital and Harvard Medical School/NINDS, UC Irvine/Flickr (CC BY-NC-ND)


