It’s been called the forgotten pandemic, yet it happened less than a century ago. Many people are dimly aware of the 1918 flu pandemic, often called the Spanish flu, but few people grasp how truly consequential it was. It’s estimated to have killed 50 million people in just a few years, a number that’s sure to rise as researchers dig deeper. That’s more than were killed in battle during World War I, which was grinding to a halt as the flu was gathering steam.
Though most of us know little about the 1918 flu, it has taught scientists much about influenza, including the way the virus moves between hosts, how the human immune system responds, and how reservoir hosts like birds and pigs drive outbreaks. Perhaps most important, it has taught us how to prepare.
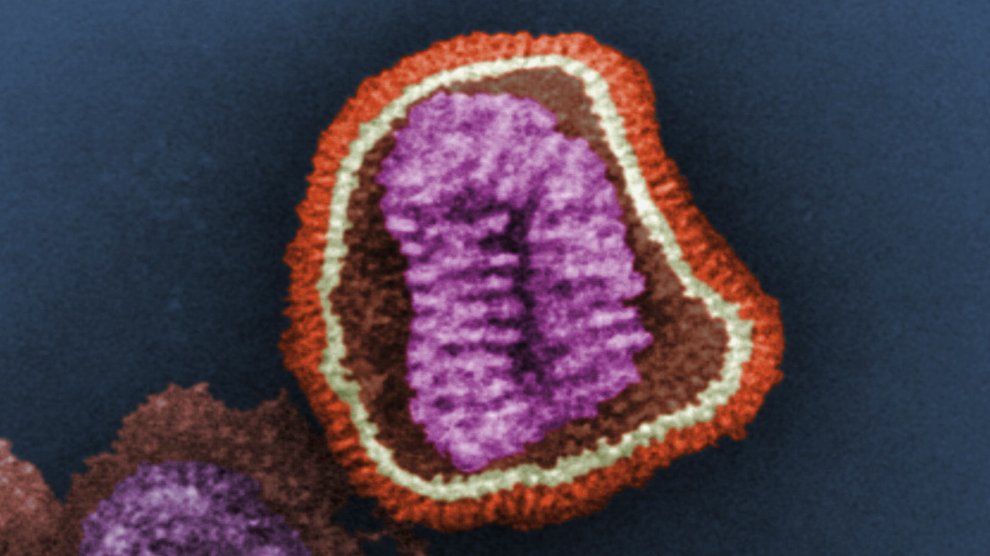
Virologists are concerned that the H7N9 strain which recently surfaced in China could be the next strain to break out of its animal-to-human transmission pattern and jump freely from person-to-person. Whether it’s H7N9 or some other strain that sparks the next widespread pandemic, virologists and public health experts figure it’s only a matter of time before a deadly influenza outbreak sweeps the globe.
Today, a handful of scientists and historians are revisiting the worst flu pandemic in recent history to help us better prepare for the next one. They’re reconstructing the 1918 flu virus, poring over historical records, and devising procedures for hospitals and the general public that—they hope—will keep the next outbreak from turning into a catastrophe. Because of what we have learned from the 1918 flu, “We understand a lot about how to plan for the next event, even how to look out for it,” says Cameron Wolfe, an infectious disease specialist at Duke Medicine.
Everything Old is New Again
Our first line of defense against influenza was also used during the 1918 flu pandemic—quarantines. Keeping the infected away from the healthy may sound overly simplistic, but it’s effective.
A few years ago, a team lead by Howard Markel, a medical historian at the University of Michigan, submitted a report to the Centers for Disease Control and Prevention about the use of “nonpharmaceutical influenza mitigation strategies,” or quarantines, in the 1918 pandemic. They found that the strategy worked well when applied appropriately. In one example, St. Louis closed its schools early in the outbreak, a move that disrupted daily life but appears to have substantially reduced mortality rates compared with other cities like Cincinnati, which waited longer. Markel and his team also found that quarantines worked at smaller scales, too. In close quarters like military barracks and college dorms, isolating patients seems to have to reduced rate of infection.
Unfortunately quarantines weren’t universally applied in 1918, likely making the pandemic worse than it could have been. This was especially so in the crowded, filthy trenches of the Western Front in World War I. There, young men, immunosuppressed by the grim and taxing conditions, frequently fell ill with the flu. But rather than instituting quarantines, the military moved the sick through an elaborate series of hospitals, exposing people along the way.
Paul Ewald, an evolutionary biologist at the University of Louisville, has studied the system closely. Once ill, “people were moved very quickly out of the trenches,” Ewald says. Their first stop was to one of many temporary hospitals on the front line, where they spent an hour or two. Next, they were moved to another group of hospitals just behind the front line. Originally constructed to treat wounded soldiers, the hospitals were overwhelmed by the flu. “These hospitals…were made to house maybe 200 people, but by 1918, they were getting 2,000 people going through a day,” Ewald says. After a brief four-hour stay at those hospitals, the infirm were loaded onto trains, “stacked like pancakes,” to a third hospital, which often had thousands of beds. “They were like big warehouses,” Ewald says.
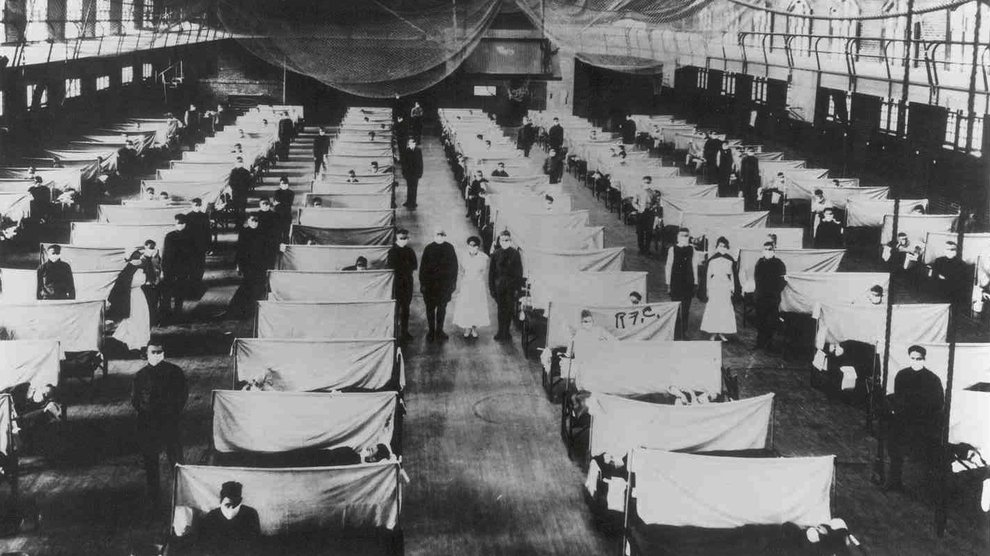
It’s a strategy that seems inspired by Ford’s then-new assembly lines. Sick soldiers were moved from one hospital to another as team after team of nurses and doctors triaged and treated, each specializing in a certain step of the process. It’s a system that works well for factories, but was devastating for public health. As patients moved through the system, they exposed dozens, even hundreds, of people to the virus. “You had the potential for individuals who were completely immobilized, sick with a nasty strain, to actually serve as a source for infecting many other individuals—even if they were on their deathbed,” Ewald notes.
The stark differences between St. Louis and the Western Front illustrate the importance—and efficacy—of quarantines. Since 1918, we have finely tuned our quarantine procedures. We quickly isolate infected patients, keeping them not only within the same hospital but often within the same room. If they need to be moved, they’re not shipped on packed railcars or crowded planes, but transported in their own ambulance, helicopter, or medical flight, minimizing others’ exposure to the virus.
Rebuilding the Virus
At the time of the 1918 pandemic, scientists and public health officials didn’t even know it was caused by a virus—the influenza virus wouldn’t be discovered until 1933. Since that time—and especially in recent years—we’ve learned much more about the germ.
Jeffrey Taubenberger is a virologist at the National Institute of Allergy and Infectious Disease, who, for most of the last 20 years, has been tracking down the molecular and genetic remnants of the 1918 flu virus. His quest began in the mid-1990s when he was working at the Armed Forces Institute of Pathology. There, he stumbled on the institution’s vast preserved tissue collection, which happened to contain around 100 samples of lung tissue from autopsies taken around the time of the pandemic. Not all of them contained fragments of the virus, but he gathered enough to paint the first genetic picture of the deadly strain.
In the years since, Taubenberger and his team have learned that the 1918 influenza strain is a marvel of virus evolution; in their experiments, it has replicated extraordinarily well. In particular, its polymerase—an enzyme that makes copies of the virus’s genetic material—is especially suited to its job. “The 1918 virus is like a really high-end sports car,” Taubenberger says. “All of its parts are just really well engineered, and they all work together very efficiently.”
Fast replicating viruses can overwhelm a patient, but the 1918 virus had another trick up its sleeve. “It some how induces this really strong inflammatory response,” Taubenberger says, turning the body’s defenses against itself. Our immune system’s actions are intended to rid our bodies of viruses and other infections, but too vigorous a response can be counterproductive, even dangerous, as was the case with the 1918 flu. “We and others think that this inflammation that’s induced by the virus might actually be playing a big part in why the virus was so virulent.”
That finding has inspired Taubenberger to investigate new ways of fighting the flu, ones that could be applicable to future influenza outbreaks. “It might be possible to treat severe influenza by modulating the immune response, not necessarily by trying to block the virus from replicating,” he says. Doing so would be a delicate balancing act. Even in cases where your immune response causes harmful inflammation, your body still needs it to clear the virus from your system and develop an immunity against it. But if Taubenberger can figure out what initially triggers a harmful immune response and limit its intensity, it could provide doctors with another tool in case of an outbreak.
Global Surveillance
Perhaps the lesson with the broadest impact is that of international cooperation. In 1920, with echoes of the pandemic still reverberating, the League of Nations created an internatinal health organization. Though influenza wasn’t its only focus, the 1918 flu “punctuated the need for greater collective action,” writes Kelley Lee, a professor of global health policy at the London School of Hygiene and Tropical Medicine, in her book on the World Health Organization.
As an entity, the LNHO didn’t last long. Bureaucratic wrangling between it, the Paris-based Office International d’Hygiène Publique, and the United States Public Health Service limited the organization’s effectiveness. The start of World War II finished it off. But the LNHO was still an important step. Countries became more accustomed to sharing information, something that is vital when dealing with a fast-moving epidemic. (A classic case of misinformation comes from the 1918 flu, which is often called the Spanish flu. During World War I, most countries censored their news to prevent leaking information to the enemy. But Spain wasn’t involved in the hostilities and didn’t censor its health reports. People assumed it started there.)
The LNHO would foreshadow the later establishment of the World Health Organization, also known as the WHO, which today plays a key role in monitoring flu outbreaks and coordinating response around the globe. “The World Health Organization, in the last few decades, has actually put together a really fantastic influenza surveillance network globally,” Taubenberger says.
Be Prepared
It’s been nearly a century since the 1918 flu swept the globe, and while popular culture has largely forgotten about it, the medical and public health communities haven’t. We’re fortunate that the pandemic coincided with rising sophistication in the disciplines of medicine, epidemiology, and public health. We’ve taken the lessons learned from that time and honed them on subsequent outbreaks.
Because of that, the United States and other developed countries are particularly well prepared, notes Wolfe, the infectious disease specialist. “We have large plans already drawn up, stockpiles drawn up, worst case scenario plans for [answering], ‘How do we keep critical services flowing in the event of a large burden of patients?’ ” The trick will be spreading that know-how to developing countries.
The sooner we mull those plans over, the better. “The more you explore these planning issues, the more you dig up other problems,” Wolfe says. “Hospitals in U.S. are fantastic at keeping themselves at capacity,” he says. “We do a great job at making sure we’re always full. So you then say, how would your healthcare system work if we suddenly got 200 extra people who needed ventilators? What would we do in that situation?”
Another missing piece of the puzzle, Taubenberger says, is better influenza monitoring in animal populations. “It is very important to expand surveillance into animals,” he says. Doing so can give us earlier intelligence about which strains may be prevalent in the coming flu season, enabling drug makers to target their vaccines more effectively and giving public health officials enough advance warning to plan appropriately.
Those and other challenges are why Taubenberger, Wolfe, and others have stayed on the case. “The reason that we study 1918 is not just out of historic curiosity,” Taubenberger says. “I feel very strongly that if we continue to understand the lessons of 1918, to push us to ask and answer these questions, it’s only going to help us prepare and hopefully be able to better respond and treat influenza in the future, even if we’re not quite there yet. The story is still unfolding.”


