Finding Alternatives to Opioids
As the opioid crisis continues to unfold, doctors and scientists are exploring other ways of fighting pain.

The young cook had just turned away from the deep fryer when he heard a thud and felt a searing pain that stopped him in his tracks. “It was so intense that my whole body kind of froze for a minute,” recalls Kevin Walsh, propped up in a hospital bed, nine days later. “It was just so, so excruciating.” An aerosol can of non-stick spray that somehow fell into the fryer had just exploded, blowing a cascade of burning oil across Walsh’s back. Two co-workers were burned as well.
Walsh is telling the story in the burn unit at Harborview Medical Center in Seattle. A few hours from now, he’ll face debridement, a procedure that can rival the initial injury in terms of producing pain. Every day, nurses change his dressings and clean his wounds, scraping away dead tissue from tender, inflamed nerve endings.

It’s a miserable experience, but Walsh’s last session was tempered by something surprising: an immersive video game called “SnowWorld.” As nurses scrubbed at his wounds, Walsh wore a headset and tried to pelt attacking penguins with snowballs. Despite simple graphics that pale in comparison to his beloved video game “Call of Duty,” Walsh says, “I have to admit, it did offer a distraction. I could tell they’re peeling off a bandage and I remember actually thinking in my head, ‘You know this should hurt a bit more,’ but I was focused on the game, so it didn’t. I was distracted.”
SnowWorld was developed under the direction of Dr. Hunter Hoffman, a research scientist in medical engineering at the University of Washington School of Medicine in Seattle. Over the past two decades, he and his colleague Dr. David Patterson, a psychologist, have pioneered the technique of using immersive virtual reality (VR) to distract people during painful medical procedures.
VR is being incorporated into a variety of medical fields—including treatment for post-traumatic stress disorder —and is now being used to control pain. “Logic says that pain requires attention, and for some reason, going into the computer world takes a lot of attentional resources,” Hoffman explains. Your mind can’t be focused on pain and something else at the same time.
SnowWorld and its cruder predecessors have been used by hundreds of burn patients at Harborview and at Shriners Hospital for Children in Galveston, Texas. Hoffman and Patterson also conduct controlled experiments with healthy volunteers in which “pain” is caused by a small, metal device that delivers heat at carefully calibrated levels. The volunteer subjects control the intensity; Hoffman and colleagues set an initial pain level and then measure how it’s affected by various interventions. With VR, the pain is reduced by 30–50%.
Brain scans performed using fMRI illustrate the biological underpinnings. Under painful stimulation, Hoffman says, at least five distinct areas of the brain typically light up, reflecting the complexity of the experience. The two most relevant are the anterior cingulate cortex, associated with the emotions that come with pain, and the sensory cortex, channeling the pain’s intensity. Brain scans show that VR can reduce the intensity of pain by 30% or more.
When it comes to treating pain, VR may be the tip of the iceberg. Doctors have been exploring everything from cognitive behavioral therapy and new forms of non-addictive painkillers to yoga, meditation, and even hypnosis. While the ultimate potential of each approach remains to be seen, it’s clear that opioids are just one—very flawed—way of dealing with pain.
A Complex Target
The way most of us conceive of pain is pretty basic: hammer hits thumb. Nerve endings send alarm signal up the spinal cord to the brain. We recognize the signal as pain, and respond, ”Eeyow!” But as you’ve probably guessed by now, it’s a bit more nuanced than that.
“When pain persists, it becomes far more complicated, because the experience is perceived throughout the brain,” says Dr. David Tauben, who heads the Division of Pain Medicine at the University of Washington School of Medicine, which oversees Harborview.
“The brain doesn’t like silence, the absence of a signal.”
The classic example is phantom limb syndrome. Patients who suffer an amputation frequently feel sensation, often painful, where the limb used to be. According to Tauben, some of that may be due to damaged nerve endings that are constantly firing. But a bigger factor is inside the skull. “The brain doesn’t like silence, the absence of a signal,” he says. “It’s not a normal state, and so it fills the void.” A common treatment is mirror therapy, in which the patient uses a simple device to hide her missing appendage and looks into a mirror, which seems to reflect two healthy limbs. Repeating the simple trick often leads to improvement.
Pain’s complexity begins even before signals reach the brain. The “Gate Theory,” established by Ronald Melzack and Patrick Wall in the mid-1960s, suggests that the initial stimulus of pain is mediated by a “gateway” of special cells in the spinal cord. These cells interpret that stimulus as pain or as something else. The interpretation is constantly being modified by new sensory inputs as well as by signals coming from the brain itself.
Two anecdotes paint a vivid picture of how dramatically our experience of pain is shaped by other sensory inputs, including our expectations and our “rational” thought process. Lorimer Moseley, a professor of clinical neuroscience at the University of South Australia, tells the story of being bitten by an Eastern Brown snake while hiking; its bite is reputed to be one of the most painful things in the world. Moseley, however, didn’t see the snake and thought he’d simply brushed a twig. He felt little pain, right up to the point where he passed out and nearly died. On a subsequent hike, when Moseley was in fact pricked by a twig, he says he felt it as searing pain.
Another account, published in British Medical Journal , describes a construction worker who was rushed to the hospital in excruciating pain, after he jumped from a roof and saw a thick, 6-inch nail pierce his foot. When it was removed, it became apparent that the nail had gone through the boot but passed between his toes. The pain evaporated. As the attending doctor wrote: “A marvelous cure appeared to have taken place.”
Need for Swift Intervention
The power of the mind is even greater, Tauben says, in cases of chronic pain, which is defined as pain which lasts more than 90 days. “Somewhere between day five after a trauma and day 90, the central nervous system becomes transformed,” he says. The pain is no longer being generated at the site where it began. It’s a constant, reverberating loop, like a stuck car alarm.
“Most cases of chronic pain are complex,” says Dr. Helene Langevin, director of the Osher Center for Integrative Medicine at Brigham & Women’s Hospital in Boston. “The reaction of the central nervous system can amplify and perpetuate the pain.”
Most doctors agree that it’s crucial to try and prevent chronic pain from taking hold in the first place. Treating pain after surgery or injury doesn’t just bring relief in the moment. It prevents the nervous system from laying down new pathways that can become permanent.
Chronic pain afflicts tens of millions of Americans, although the precise figure is hard to pin down and somewhat controversial. What’s clear is that the burden is enormous. A 2011 report funded by the Institute of Medicine found the cost of treating pain exceeds what we spend on care for heart disease and cancer combined. That money has also fueled the ongoing and devastating epidemic of opioid abuse. As the toll of addiction grows, doctors are seeking, and finding, fresh alternatives.
Hypnosis
SnowWorld, Hoffman and Patterson’s VR program, was developed to distract the mind during periods of intense, predictable pain like the debridement of burn patients. Another patient used SnowWorld to successfully blunt the pain that flared every time he removed his leg prosthesis. Now, Patterson is also exploring whether a different model of VR—a form of hypnosis—can combat the non-stop nagging of chronic pain.
His first stab at hypnosis in the hospital was an act of desperation. An older burn patient, in agony, refused to have his dressing changed—so Patterson read a hypnosis script that ended with the instruction, “When the nurse taps you on the shoulder, you will feel nothing but comfort.” The tap came, the patient fell asleep and he stayed zonked until his wound care session was done. Patterson was hooked.
Today, Tim Clark, a 69 year-old retired construction project manager, is at Harborview for his regular session. Sitting in a wheelchair, he stares at Patterson and listens to his low, soothing incantation.
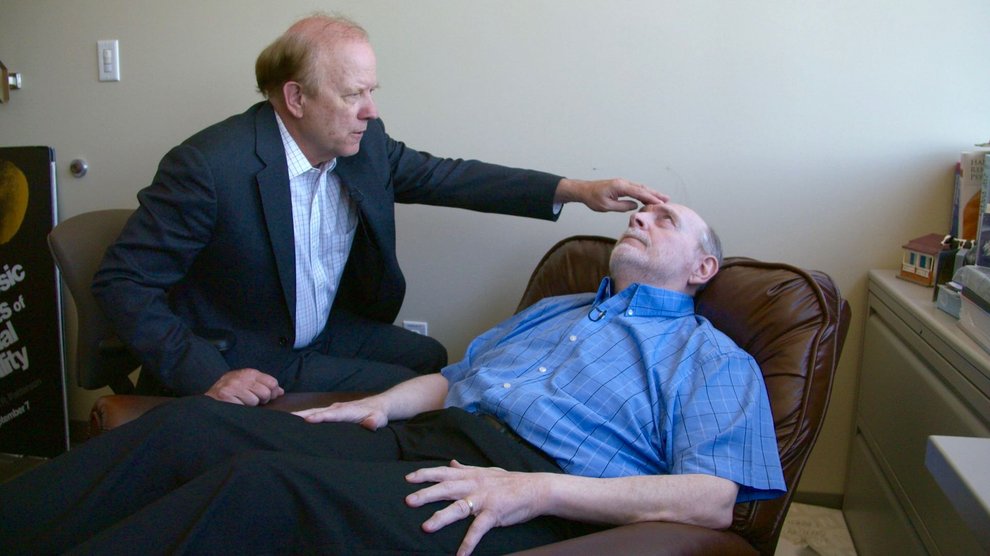
Clark’s story is tough to hear. Sitting at his desk one day, he felt a sudden stabbing pain in his back. Within two weeks, nearly his entire body was paralyzed. He was diagnosed with Guillan-Barre syndrome, a neurological disorder, “One day and my life changed forever,” he says.
Work was impossible. Clark’s adult daughter, a college graduate, moved home to help care for him. Nerve damage led to severe back problems and three surgeries to fuse the vertebrae. Over time, the pain became unbearable, including episodes that left Clark barely able to move for up to 15 hours at a stretch. “The most frustrating part is that I can’t anticipate it. I can be perfectly fine and then five minutes later, I’m getting these shocks in my nerves. It’s like short circuits, firing away. At times I can’t even touch my skin, it’s so painful.”
Heavy doses of opioids left Clark “in a real funk.” Then, he met Patterson. “As soon as we started doing hypnosis, it was evident that this was going to be valuable. It was going to give me more tools to deal with the pain instead of just grabbing pills.”
Clark still uses opioids. He wears a fentanyl patch, and takes an occasional Dilaudid to control flare-ups, but he’s down from three or four pills a day, to three or four a week. Instead, he relies on hypnosis, which he likens to meditation. “The brain can’t think of two things at the same time. If I’m focusing on the pain, then everything else stops. If I focus on something else, the pain doesn’t go away, but it diminishes.”
Along with sessions here, Patterson has recorded half a dozen versions that Clark listens to at home. “I practice pretty much every night when I’m going to sleep,” he says. “It brings me to the present, and I’m not so worried about my pain. It’s just dealing with what’s there right now. I spent a lot of years in competitive karate and I’ve always believed in mind-over-matter stuff. I just focus on something other than the pain.”
Mind Over Matter
Tauben likes to recall an example from the waning days of World War II, when the Allies were storming Anzio Beach in Italy. Dr. Henry Beecher , a pioneering anesthesiologist, interviewed 215 combat troops at the scene and reported that most, even those with serious injuries, were in so little pain that they turned down morphine. Whether it was due to the stress of being in a war zone or relief at being rescued, Beecher couldn’t say, but he concluded, “Strong emotion can block pain. That is common experience.”
Intense stress or danger triggers an array of physical responses, including the release of hormones including endorphins, adrenaline and cortisol—the so-called stress hormone—all of which play a role in suppressing pain, Langevin says. “These things keep us going in the face of a threat. If you’re running from a lion in fear of your life, you’re not going to worry about stepping on a sharp piece of rock.”
To Tauben, the lesson is broader. “A major approach is finding meaning and purpose in your life. If pain is the only thing that matters, it becomes the most important thing.”
Four-thousand miles and seven decades from Anzio Beach, a 32-year-old writer named Dustin Grinnell can attest to that. In late 2015, Grinnell ditched a corporate job in Boston and hit the road, riding his motorcycle across the country to try his hand at becoming a full-time screenwriter. Within a few months, the dream had gone sour. The work wasn’t coming. There were tensions with roommates, he and his girlfriend broke up, and then Grinnell tweaked his back while jogging
Looking back, he sounds rueful. “If I’d been in another job or had good health care, I probably wouldn’t have thought much of it. But with the vulnerability of my position—financially and even romantically—I began to fixate on the pain.” A bad week of flare-ups, he says, “scared the sh– out of me. I sort of catastrophized, and I became overwhelmed with panic and stress.” He says the injury, coupled with depression, changed his whole self-image. “You’re hobbled, you’re no longer whole. I started to think: ‘I won’t run again; I can deal with that, but can I work? Can I sit? Can I write?’”
X-rays and MRIs didn’t find any structural problems that could explain his suffering, but Grinnell only found relief after he started on antidepressants. He took a new doctor’s suggestion to get his body moving and to try an eight-week course in “mindfulness-based stress reduction.” “Now I swim a couple of times a week. I do yoga. I can lift weights. With exercise, medication, meditation, and psychotherapy, I climbed out of the whole thing.”

Dr. Robert Jamison, a pain specialist at Brigham and Women’s Hospital, where Grinnell landed a job as a copywriter a few months ago, says the story doesn’t surprise him. “People who are depressed and anxious just don’t cope as well as people without a mood disorder.”
Some bristle at the notion, “it’s all in your head,” but doctors are finding success with a variety of approaches that have a strong psychological component. Cognitive-behavioral therapy alone has been found to reduce pain. Other experiments show that simply being physically close to someone who seems sympathetic can reduce pain’s intensity. It’s likewise reduced when doctors explain what to expect in coming hours or days.
This spring, the American College of Physicians issued new guidelines for the management of chronic low back pain, including a “strong recommendation” that patients try mindfulness-based stress reduction or other alternative therapies before turning to medication.
Langevin says it’s important to first determine whether the underlying cause of pain is still there, and if it’s treatable. “It may well be that something is actually wrong with tissues, not just the nervous system.”
Her research is focused on the role of connective tissue in chronic pain and on treatments that use movement or manipulation to relieve it. At the Osher Center, patients may undergo yoga, acupuncture, or massage along with mindfulness-based therapies and more conventional treatments. “The treatments are integrative. It’s not one approach,” Langevin says.
Another Option
Yoga and meditation aren’t the only “alternative” treatments to have received a mainstream stamp of approval. A January report from the National Academies of Science, Engineering, and Medicine found strong evidence that cannabis—known to many as marijuana—reduces pain, especially neuropathic pain, a complex form of chronic pain that typically involves damaged nerve endings. Patients weren’t waiting for the news; doctors who work with cannabis already say the vast majority of patients who come in the door list pain as their primary complaint.
Dr. Jordan Tishler, an internist who spent 15 years handling emergency medicine at the Boston VA, last year launched a cannabis-centered practice with two locations just outside Boston. He says many patients are looking to reduce their use of opioids, and he’s glad to offer something he says is safer.
One of his patients is Harris Barron, a 90-year-old artist, World War II veteran, and retired teacher with rheumatoid arthritis. He avoids opioids. he says, because “I don’t like being doped up all day. I like a clear head.” Since starting to use cannabis about a year ago—typically once a day before bedtime—he’s cut his daily intake of ibuprofen by about 40%. Barron still senses the pain of arthritis, but says it’s no longer disabling or all-consuming.
Opioids mimic natural brain chemicals, but with critical differences.
Tishler adds that most patients do well with low doses of cannabis. “The amount they find relief with is nowhere near what laypeople recommend to each other or what recreational users take.”
In a sense, the brain comes built to respond to cannabinoids like THC and CBD, key chemicals in marijuana. In fact, the brain makes its own so-called endogenous cannabinoids, which fit the same receptors the way a key fits a lock.
Opioids also mimic natural brain chemicals, but with critical differences. Opioids are taken up by receptors that control our breathing; filling too many receptors leads to a potentially lethal overdose. In stark contrast, cannabinoids don’t affect the brain stem, where breathing and other basic life support functions are found. As a result, you can’t fatally overdose.
Some studies suggest that states where patients can legally use cannabis have lower rates of opioid addiction. But no clinical trial has tracked individuals to see if cannabis helps reduce their opioid use. Dr. Timothy Naimi, an internist and public health specialist at Boston University School of Medicine, says it’s too early to draw conclusions. “In the state-level studies, we don’t even know how many folks were using marijuana already.”
Naimi also worries that cannabis use might increase the number of accidents—the data is inconclusive—and that some new users will become dependent.
Treatment of Last Resort
The explosion of opioid use came after the publication of studies which found that terminal cancer patients were able to use the drugs without developing the kind of dependence and negative side effects that many predicted. Helped by a marketing push, the drugs were soon being prescribed for a variety of long-term pain patients.
It became apparent that the sense of security was false, and rates of addiction began to surge. At the same time, recent literature reviews find that for most chronic pain, opioids offer only a modest benefit.
Even so, no newer approach can rival opioids when it comes to providing immediate relief from severe, acute pain. Kevin Walsh, the burn patient, was still gushing over VR and video games when he recalled one of his first wound care sessions. “They started to do it, and the pain was just unbearable. And so they gave me an injection of fentanyl in my IV, and minutes later, I felt almost nothing. The pain was gone.”
Look for the PBS NewsHour report about pain treatments on the Leading Edge of science, October 4, part of a week-long series about opioids.
Photo credits: Caleb Hellerman, frankieleon/Flickr (CC BY)


