A new form of carbon is born—on a bed of salt
The long-sought molecule could one day power high-energy electronics.

An artistic three-dimensional representation cyclo[18]carbon, a ring of 18 carbon molecules. Image Credit: IBM Research
A new molecule can now officially join the ranks of Club Carbon.
The chemical debutante is cyclo[18]carbon, a ring of 18 carbon atoms that’s finally been created, imaged, and analyzed in a report published today in the journal Science. The findings help settle decades of debate on the structure and properties of the molecule, which some researchers doubted could even be stably made in the lab.
Carbon is well-known for its versatility. Even without input from other elements, atoms of carbon can link up to create a diverse set of compounds, ranging from hard, clear diamonds to malleable black graphite. Without much prompting, carbon can come together in thin, flat sheets, or intricate, three-dimensional crystals; it can arrange itself into meticulously structured lattices, or slouch into amorphous blobs.

A model of cyclo[18]carbon's molecular structure. Image Credit: IBM Research.
But one structure eluded scientists for decades: an elegant ring of carbons, unsupported by any other stabilizing element. Previous attempts to generate so-called cyclocarbons in the lab had resulted in volatile gas, too ephemeral to study in any rigorous way.
The recent breakthrough, however, shows that a cyclocarbon can be generated by pruning carbon and oxygen atoms off a more complex molecule at extremely cold temperatures with a technique called atomic force microscopy. Starting with a molecule made up of 24 carbons linked to six oxygens, researchers at IBM Research and Oxford University were able to generate a final structure of 18 carbons—the smallest possible ring of carbons predicted to be stable by theoretical models. Imaging and further analysis also revealed that the atoms in the cyclocarbon were joined together by alternating single and triple bonds, rather than a uniform chain of double bonds.
It was a harrowing process—the culmination of many months of trial and error, study author Lorel Scriven of Oxford University told Ryan F. Mandelbaum at Gizmodo. The actual synthesis of cyclo[18]carbon was only part of the battle: To keep the unstable molecule from reacting into other forms of carbon, or creating a hybrid element with something in its surroundings, the team had to conduct their experiment on a bed of inert table salt.
But cyclo[18]carbon’s fickle nature is part of what makes it potentially powerful, too. “At some point we might be able to controllably trigger such reactions and create more complex molecular structures at will,” study author Katharina Kaiser, a physicist at IBM Research, told John Wenz at Popular Mechanics. This could give chemists an unprecedented amount of control over the synthesis of carbon-based molecules.
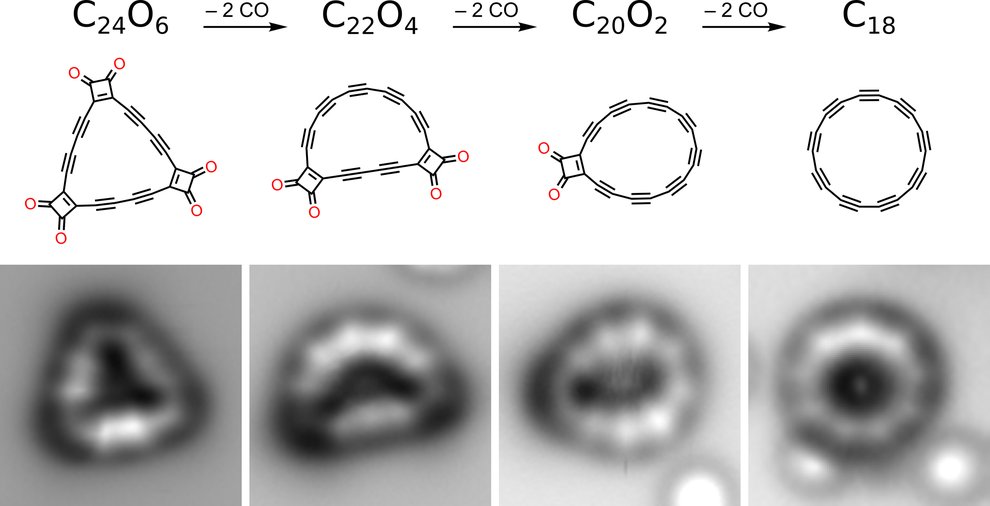
The process by which cyclo[18]carbon was synthesized in a new study. C24O6, a molecule with 24 carbon atoms and 6 oxygen atoms, was pared down into a ring of 18 carbon atoms. Image Credit: IBM Research
It’s not totally clear what’s next for cyclo[18]carbon. But if it’s anything like graphene—another form of carbon that’s extremely good at conducting electricity—the new molecule could someday play a role in efficiently powering devices that require a lot of processing power, like those relying on artificial intelligence, Wenz reports.
Regardless of what the future holds, simply locking down cyclo[18]carbon is a big win for the chemistry community.
As chemist Rik Tykwinski of the University of Alberta in Edmonton, Canada, who was not involved with the research, told Emily Conover at Science News, “It’s not every day that you make a new form of carbon.”



