Do Genes Dictate How Mice Cope With Trauma?
Scientists have uncovered a gene that affects how mice encode fear-based memories—which could inform the study of psychiatric disorders like PTSD in humans.

A gene called TIA1 might play a big role in determining how female mice remember stressful and scary experiences. Image Credit: zsv3207, iStock
For better or worse, memory operates most potently at emotional extremes. Happy memories often tend to stick; so, too, do some of the ones we’d most like to forget.
Much of how the brain selectively consolidates such events remains mysterious. But today, in a study published in the journal Cell Reports, a team of researchers takes the first steps towards pulling back the veil on how a gene called TIA1 might control fear-based memories in mice. If confirmed in humans, the findings could someday have implications for understanding stress-related psychiatric conditions such as post-traumatic stress disorder (PTSD).
Animals’ ability to remember impactful experiences is arguably one of our greatest assets in the quest for survival. In humans, a good memory is what keeps us afloat, from maintaining to-do lists to giving us pause before ingesting spoiled foods. But memory can also become a double-edged sword—especially when recollections of a traumatic experience become intrusive or debilitating, and start to interfere with daily life. Occasionally, this can manifest in psychiatric illness like PTSD.
Not all trauma sticks with us, however, and the same traumas don’t affect individuals in the same way. For years, researchers have been working to uncover what aspects of our biology underlie these differences, and how vulnerable individuals can be identified and supported. “Environment plays a big role in psychiatric disorders like PTSD,” says Rachel Yehuda, a neurobiologist and PTSD expert at the Icahn School of Medicine at Mount Sinai who was not involved in the study. “But there’s always been this nagging question about why people respond differently to adversity.”
To untangle these dynamics in mice, a team led Joseph Rayman and Eric Kandel, neuroscientists at Columbia University’s Zuckerman Institute, focused their efforts on TIA1, a protein that’s abundant in the hippocampus, the memory consolidation center of the brain. Previous work from Kandel’s group had shown that TIA1 helps cells manage stress in their environment, but whether this translated up to the level of behavior remained unclear. So the researchers bred male and female mice that lacked the gene that encodes TIA1 and put their stress management skills to the test.
A typical mouse doesn’t think much of the scent of ethanol. But if that smell is wafted in a rodent’s direction while tiny electric shocks are being administered to its feet, it will learn to avoid areas that reek of the otherwise innocuous odor—a clear indicator that it has crystallized a fear-based memory.
When male mice were taught this association, all of them shied away from chambers that bore the scent of alcohol, regardless of their TIA1 status. But to Rayman’s surprise, female mice producing normal amounts of TIA1 didn’t take the same pains to avoid the odor. It seemed that while the females remembered the scent of alcohol, and the distressing foot shocks that often came with it, the association simply wasn’t as bothersome.
In females in which the TIA1 had been snipped out, however, the pendulum swung in the opposite direction: These mice were far more likely to recoil from the odious scent.
Given the myriad differences between the hormones and physiology in biological males and females, there’s probably not one single explanation for this discrepancy, Rayman says. But one discrepancy stood out to the researchers: Female mice naturally produce more TIA1 than males. This could help explain why the females seemed a bit more resilient to stress in the first place—and why switching the gene off entirely constituted a much more striking change.
If that was the case, the researchers reasoned, then upping, rather than damping down, the production of TIA1 in male mice might extinguish some of the effects of fearful memories. And that’s exactly what they found: Male mice bred to churn out gobs of TIA1 were far less likely to shy away from the scent of ethanol.
These results illustrate two things, Rayman explains. First, TIA1’s role in forming stressful memories seems to operate on a sliding scale—meaning the amount of TIA1 an individual produces could have bearing on how they internalize difficult experiences. Second, the differences between the sexes could “shed some insight into what makes male and female brains different when processing fear memories,” he says. This latter point is especially intriguing, he says, given that both PTSD and anxiety disorders “afflict women at a far greater incidence and with greater potency.”
Of course, mice learning to be wary of a specific smell isn’t the same as human trauma. But Rayman argues that “among all psychiatric illnesses, PTSD is profitably studied in mice. It’s a disorder of fear and memory, and those are highly evolutionarily conserved.” The researchers are now conducting preliminary studies in humans, and so far, the results hint that TIA1 functions similarly across species, Rayman says.
Still, “this doesn’t guarantee that [TIA1] will be associated with human PTSD—that’s a big leap,” Yehuda, the Mount Sinai neurobiologist, says. “But it’s an impossible leap without having some animal basis.”
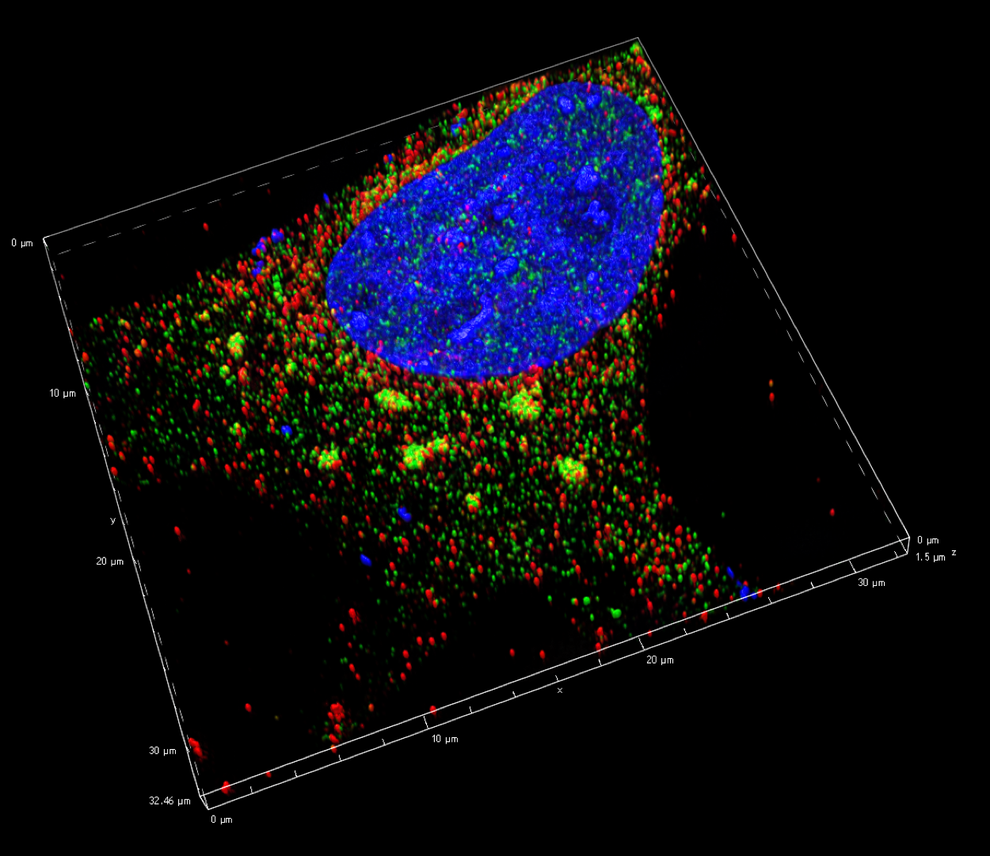
A cell showing TIA1 in yellow and green. TIA1 helps cells deal with stress by sequestering non-essential molecules so resources can be diverted to deal with the situation at hand. Image Credit: Joseph Rayman, Kandel Lab, Columbia University’s Zuckerman Institute
Additionally, more work is needed to fully understand exactly what TIA1 is doing at the cellular level, and how those activities might ultimate influence memory. It’s also too early to say if there’s an “ideal” amount of TIA1 for a well-functioning brain. And that answer, if it exists, might vary by biological sex or an individual’s genetic background. Still, this work points to the fact that this protein—formerly understood as an cellular asset for enduring molecular stress—can also govern biology at the level of behavior, Rayman says.
“These authors got it right,” Yehuda says. “When it comes to translating research [from mouse to human], translation isn’t transliteration. But this is an elegant piece of science that may help explain some aspect of individual difference when it comes to avoiding fear cues.”
This discovery “hits at a time when we are beginning to fully appreciate the complexity” of the relationship between brain and behavior, says Ann Rasmusson, a psychiatrist at Boston University who was not involved in the study.
Many genes factor into psychiatric illness—a fact that’s precluded a one-size-fits-all solution when it comes to diagnosis or treatment. TIA1 is likely to be only one thread in the very complex tapestry that weaves together emotion and memory. But every new factor identified could constitute critical intel, especially for the patients for whom all standard therapies seem to fail.
At the same time, that also means that a single “risky” gene is by no means a guarantee of illness or adversity. “Genes are an important piece of the puzzle, but there are so many other systems involved, and you may well have something to compensate,” Rasmusson says. “That’s something that should also give us hope.”