The Evolving Flu
The new swine flu that emerged in early 2009 and is now circulating around the planet descends from the 1918 "Spanish" flu, which killed up to 50 million people worldwide. That fact frightens many people, but for influenza expert Dr. Peter Palese of the Mt. Sinai School of Medicine, it's one of several reasons that he feels we should not greatly fear this new so-called H1N1 flu—even as we sensibly prepare ourselves for its next wave.
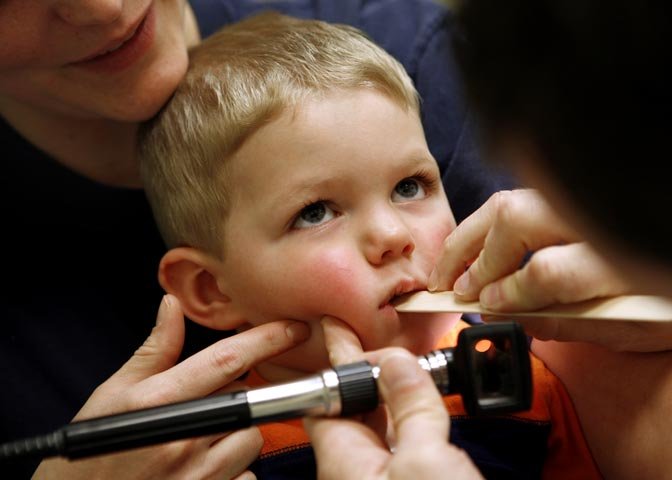
NOT SO SCARY
NOVA: Back in May you wrote an article for the Wall Street Journal entitled "Why Swine Flu Isn't So Scary." Do you still hold that position?
Peter Palese: Yes, I do. This H1N1 swine virus has properties that are clearly of concern, because it can be transmitted easily from human to human. However, it does not cause as much morbidity and mortality—as much disease and death—as other influenza viruses by virtue of its not having a particular virulence gene. Also, the human population has what we call "herd immunity" against H1N1 viruses, because these H1N1 viruses have been circulating for some time over the last century. And we still have regular seasonal H1N1 influenza viruses.
Third, [in this novel H1N1 strain] we also have a virus that is sensitive to antiviral drugs, to the neuraminidase inhibitors. [Editor's note: Neuraminidase inhibitors are a class of antiviral drugs that block the function of a flu virus's neuraminidase protein, thereby preventing the virus from reproducing.] So that gives us another tool in terms of preventing transmission and also treating patients. This is something we did not have in 1918. Also, the virus in 1918 was causing a lot of secondary bacterial infections—now we have antibiotics. Thus, this is less of a concern than it was in 1918 or even 1957 [when another flu pandemic occurred].
So I believe that we have a milder influenza virus at hand, and we also have better tools to deal with a pandemic than we had just 50 years ago.
You've said that, unlike with other flus, being older is actually a benefit when it comes to this one. Why is that?
The novel H1N1 virus is actually a descendant of the 1918 virus, and the current seasonal H1N1 virus is also descended from the 1918 virus. They are all related. Someone who is older has simply been infected more often with H1N1 virus and therefore has built up what we call cross-reactive immunity. This person is therefore better protected against the novel H1N1 virus than people who are younger and who have not experienced as many H1N1 infections.

DEFINING THE SWINE FLU
How would you describe H1N1 scientifically yet for the layman, in a brief way?
Influenza viruses are characterized by a surface protein known as hemagglutinin. Hemagglutinin comes in 16 different flavors, and H1 is one of them. The H1 swine flu virus belongs to that subgroup #1.
This strain combines genes from swine, bird, and human flus, right? How does that happen?
Yes, this novel H1N1 virus is actually a composite that received many different genes. An influenza virus has eight different RNA segments, and this particular novel H1N1 virus has received five RNA segments from a swine virus, two from a bird influenza virus, and one RNA segment from a human influenza virus. It is thus a triple reassortment or "reassortant" virus, which originated from three different influenza viruses. For that reason, this virus is quite unusual.
Evolution is very frequent and very common with respect to influenza viruses.
You said in a recent lecture that if someone had asked you what would be the next pandemic virus, you'd never have picked this particular strain with its constituents. Why?
The reason is that since 1918 we—that is, the human population—have experienced infections with H1N1 viruses. These H1 viruses have been circulating for 90 years now. So we thought that a different virus with a different hemagglutinin subtype would be the next pandemic strain, rather than an unusual H1N1 virus.

EVOLUTION IN ACTION
These viruses are constantly evolving. What are the mechanisms or strategies they use to do so?
Viruses come in all different forms. There are viruses that have DNA as their genetic information, and viruses that have RNA as their genetic information. And all viruses can change, and they do this by mutations. These mutations accumulate, and usually viruses are selected that can escape a certain immune or environmental pressure. So the fact that all viruses can accumulate mutations makes them subject to evolution.
In the case of influenza, we're dealing with an RNA-containing virus that has a higher mutation rate than many DNA-containing viruses. This high mutation rate allows the virus to escape a lot of pressures, and thus evolution is very frequent and very common with respect to influenza viruses.
It's all about survival, right, the way the virus manages to survive our immune system?
When it amplifies, the virus amplifies many different mutants. So it is not exactly the same virus that was infecting the first cell. There will be thousands or millions of viruses in there that are slightly different. So if the host infected by this virus has an immune response made against an earlier infection, then this new virus will be suppressed. However, there may be some mutants in there that look different, and the immune response of that host will not be able to neutralize that virus. That mutant will survive and will therefore take over and possibly even kill that host.
So the presence of many different mutants in the light of antibodies that are neutralizing this virus may give a virus that has many mutants a better chance to survive. This is an advantage for the virus. But also, if the virus makes too many mutants, then obviously it will not survive very well, because many mutants are not able to survive.
How fast do H1N1-type viruses evolve compared to other viruses? Are they particularly quick? Does H1N1 virus change more rapidly than, say, H3N2 influenza viruses?
I don't think there will be any difference. The rate of evolution is more or less the same for all influenza viruses. I don't think that there is an inherent difference between an H1N1, H3N2, or H5N1 influenza virus.
The rate may change in different hosts, however. In other words, in humans, because we have a very long life span and therefore we accumulate a lot of antibody and immune responses, if an influenza virus infects a human, then the virus is forced to be selected against the immune pressure. And therefore virus evolutionary rates can be much faster in humans. In a bird, which lives only a very short time, then the virus infects and the bird dies before that bird would be infected a second time. So while the mutation rate is the same, the evolutionary rate may be different, depending on in which host the virus replicates.
The measles vaccine has been basically unchanged for more than 50 years, suggesting that the virus itself has remained largely unchanged. Why do some viruses change more rapidly than others?
This is a very good question. Why is measles virus more or less unchanged? Why can we still use the same vaccine 50 years later? One explanation, and it's not a really convincing one, is that the virus simply doesn't tolerate these mutations. In other words, the architecture of measles virus is such that it doesn't tolerate many mutations—it would not be a measles virus anymore. Whereas influenza viruses, by virtue of, let's say, having a more flexible outside coat, may be more tolerant of these changes, and the virus takes advantage of that. By changing its outside, it evades the immune system and can therefore reinfect much more readily than a measles virus, which cannot change as effectively.
GENE SWAPPING
As you alluded to earlier, viruses also "evolve" by swapping genes with each other.
Yes. Influenza virus, by virtue of having a segmented genome, can change not only by mutations that allow it to evolve, but it can also exchange these RNA segments by acquiring such a segment from a different influenza virus, and thereby change rapidly. This is also evolution, but it is evolution that is much more rapid and allows the virus to change much more dramatically.
Humans also give viruses to animals—to pigs, to birds. It is not a one-way street.
How easy is it for viruses to swap chromosomes? Is this a rare or common event?
The event of reassortment can take place if two different influenza viruses infect the same cell. That cell can be in humans, in a pig, or even in a bird. And when this happens, these two viruses amplify—propagate, multiply—their RNA segments, and then viruses can emerge that have segments from either parent. That can happen, and is actually observed, quite frequently.
Not all of these resulting viruses are fit to survive and are easily transmitted. Thus, we don't see many of these reassortant viruses making it into the human population. But the event, per se, is probably very frequent.
With so many people getting the swine flu at the moment, is it a scary proposition that this kind of swapping of genes could happen each time it enters a new host?
The new H1N1 virus has already undergone this gene swapping, and it is possible that, as it gets passaged more and more often in humans, it might again acquire another gene. However, the possibility that such a virus becomes a more virulent virus that has a survival advantage is probably not that high. So yes, these reassortments, these gene-swapping events, can occur frequently. But the viruses that emerge or result from such a chromosome exchange are not always more fit and more likely to survive in the human population. Or in an animal population for that matter.

The swine flu seems to be easily transmitted from person to person but has not been highly virulent, whereas the H5N1 bird flu of a few years ago was just the opposite—deadly but not easily transmissible. Could the two combine forces?
That's a good question: Can a virus such as the present swine virus, which is not that virulent compared to other pandemic influenza viruses, can it combine or swap genes with another more virulent influenza virus such as the H5N1 virus and thereby become a virus that is more dangerous to humans?
That is, theoretically, a possibility. However, I think there are some restrictions to an effective gene swapping. It is sort of like if you have a Mercedes and a VW— just putting the powerful Mercedes engine into a VW may not make a racing car. These genes may not fit as well, even though there is a very highly virulent engine, so to speak, just as a Mercedes or Lamborghini engine may not be optimal for a VW or Honda. So these exchanges of genetic information can happen, but the result may not always be a more effective car.
JUST ONE PROTEIN
You said at the outset that one reason the current swine flu is not as virulent as earlier pandemic flus is that it's missing a certain key protein. Is this the same sort of thing—a slight change meaning a huge difference?
Yes. The 1918 virus and the two other pandemic strains of the 20th century, the 1957 Asian flu and the 1968 Hong Kong flu, all expressed one protein, for which the lab jargon is PB1-F2. And this protein is a virulent protein; it gives the virus an extra advantage in terms of causing disease. Now, this protein, this PB1-F2, is missing in the new novel swine H1N1 influenza virus, and that makes it less likely that this virus will be a very virulent and highly potent influenza virus.
With the swine flu racing around the world, could it acquire this missing protein during its current spread?
There is always the possibility for an influenza virus to change by mutation and also to acquire by reassortment a gene that has more virulence and more pathogenicity, and thereby become more virulent. However, it may also lead to a sort of misfit of these genes in that particular virus. Then even though it has one particular virulence gene, it may still be compromised in other properties. For example, it may not pass as easily from one cell to the other. It may be transmitted from one person to the other very effectively, but it may not as easily infect one cell from another one. Therefore a virus may end up being less potent than the one that did not have this PB1-F2 gene.

JUMPING SPECIES
How does a virus jump from one species to an entirely different species, say from pigs to humans? What happens to it genetically to prepare it for an entirely new kind of host?
Influenza viruses infect many different species—birds, horses, pigs, and obviously also humans. In each case, the viruses adapt to a particular host and are more comfortable in that host. That said, influenza viruses are still sometimes able to jump from one species into the other, and one or two mutations may make this virus then comfortable in that new host.
Again, this is very unpredictable. We don't know exactly what the mutations are that allow an avian influenza virus to suddenly grow in humans. Also the other way around, because it is quite clear that humans also give viruses to animals—to pigs, to birds. It is not a one-way street. We are constantly bombarded by animal influenza viruses, but unfortunately we also give human viruses to animals, and that obviously has great consequences for agriculture.
The virus is always ahead of us by virtue of its ability to change.
Is it the same sort of adaptability that makes a virus readily transmissible from human to human?
Well, a bird virus a priori is not transmitted easily from human to human. So not only does the first step—namely, that an animal influenza virus infects a human—requires in most instances some changes. But if that virus then is to be readily transmitted from human to human, it has to acquire usually additional changes that allow that transmission to happen.
SPRING FEVER
The current swine flu struck in springtime, after the regular flu season had ended. Why did that happen, do you think?
That is clearly an anomaly. However, I should say that the number of cases was relatively low and small; it was not a really big flu year in terms of the swine influenza. It may be that this virus is more easily transmitted in warmer temperatures. We don't know that yet, but perhaps this virus has a property that allows it to be transmitted more effectively during warmer temperature. On the other hand, it may be that the epidemiology of swine viruses would fall back into its regular seasonality pattern come the next winter period.
The 1918 flu also began in the spring with a mild form that later became lethal. What are your thoughts about whether this novel H1N1 could do the same?
Well, we have heard frequently that there was a mild spring wave in 1918, which was then followed in the fall of 1918 and early in 1919 with what we call the pandemic of 1918, a much more virulent event. It has been suggested that this more or less mild wave in April and May of 2009 may be something similar to what was observed during the spring wave of 1918.
There is no direct evidence, however, that the virus that caused this mild spring wave of 1918 was indeed a precursor to the 1918 virus in the fall. This could have been a different strain; it could have been one of the old strains. I'm not convinced that there is "epidemiological" evidence from 1918 that has any real bearing on the current situation, in which a mild outbreak of the swine virus is necessarily to be followed by a more virulent phase in the fall.

DOING BATTLE
When it comes to changes, either from mutations or from gene-swapping, can we do nothing but watch and wait? Or is there a way to see them coming and head them off at the pass, as it were?
We are not in a position to predict what kind of changes will happen. We are certainly not in a position to anticipate when a new subtype will occur. The virus is always ahead of us by virtue of its ability to change and by virtue of the unpredictability of when a new reassortment will occur that will have a pandemic potential. So we are in a reactive rather than proactive mode.
Can new strains evolve, say, mid-season, catching us off guard with our vaccines that we worked hard to prepare for the new year?
It hasn't really happened that often. Sometimes we do have a choice between two or three slightly different strains. I served on the FDA [vaccine] committee for four years, and this committee makes a decision that one of, say, three strains will be the next vaccine strain. We decide in February [which to make vaccines for], and then it turns out that by November/December a different strain is actually more prevalent than the one chosen by the FDA committee.
So, yes, there are certain choices to be made. And over the last 30, 40 years, we have not always made the right decision in choosing a particular vaccine strain.
Is your lab at Mt. Sinai working on swine flu?
Yes, Mt. Sinai is one of the Designated Centers of Excellence for influenza research. There are five such NIH-funded laboratories in the country, and Mt. Sinai has one of those. We are very much involved in studying the pathogenicity and transmission of this new swine virus. We are using animal models, and we are trying to understand what this virus has in terms of making it a threat to humans, and to compare it to other influenza viruses that have been isolated over the past 90 years.


