It started with a few squiggles on a microscope slide. “I saw these little tiny structures, and initially I thought they were worms,” said Patrick Secor, a researcher at the University of Washington. Those translucent wormy-looking filaments kept appearing every single time Secor transferred bacteria from cystic fibrosis patients’ lungs into a petri dish.
Eventually, Secor and his colleagues realized that these mysterious filaments were actually specimens of a very long, very stringy virus that infects the Pseudomonas aerouginosa bacteria, a killer that colonizes cystic-fibrosis-affected lungs. That finding wasn’t a huge surprise in itself. Viruses that infect bacteria—otherwise known as “bacteriophage” or just “phage”—outnumber bacteria themselves; scientists have found them everywhere bacteria live, including human lungs. However, this particular virus wasn’t hurting its host bacteria; it was helping.
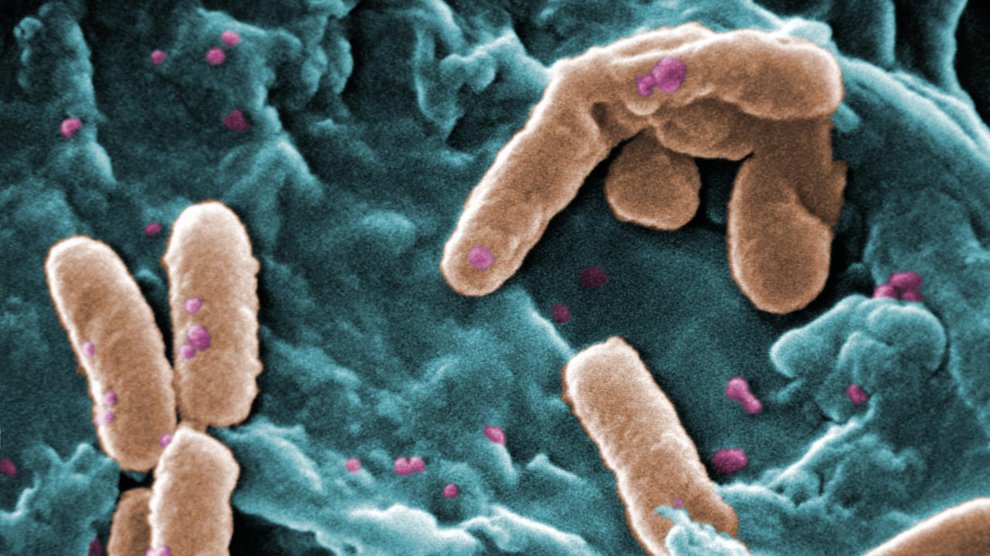
When Pseudomonas builds the phage and releases it to the environment, the phage repays the favor by creating a structure where the bacteria can live. These structures, called biofilms, serve as “housing” for both helpful and harmful bacteria, and often, when infectious bacteria colonize new territory, they try to reshape the local biochemical neighborhood to shape their needs. “Biofilms are incredibly common,” said Paul Bollyky, a co-author and infectious disease doctor at Stanford. “This ability to organize the host’s microbial [environment] into a house is really extraordinary.”
Phage filaments are especially handy for trapping free-floating bits of DNA and other biopolymers, which Pseudomonas can use for food. As the
Pseudomonas bacteria grow and divide, they spin out even more phage, creating an ever-larger clump of gunk. Eventually, in cystic fibrosis patients whose immune systems are not strong enough to kill off Pseudomonas lung infections, that gunk-clump gets so big and so sticky that the human patient can’t breathe.Cystic fibrosis patients typically have to visit a hospital every 3 to 6 months for a “clean-out”, where the doctors put them on intravenous antibiotics for a week. But even regular “clean-outs” aren’t enough. “Every night, a lot of patients have their parent pound on their back to dislodge some of the sputum,” Bollyky explained.
The co-authors hope that their finding will lead to new strategies for treating cystic fibrosis but caution that we need to learn more about the bacteria-phage relationship before we can medically sabotage it. Most methods for attacking the phage’s crystalline filaments directly would also damage human cells in the lung. It may eventually be possible to “hack” the phage’s genome so that its filaments can’t grow as quickly or form such a sticky film, but since this is the first study to pinpoint the phage as what holds the gunk together, it’s still too early to target them in patients.
Secor and Bollyky say that developing treatments for phage-formed biofilms will likely require collaboration between microbiologists and physicists who study bendable but organized materials, like the phage gunk. “Neither of us are physicists,” Bollyky said. “What happened was Pat [Secor] and I were sitting there one day, and we added a drop of phage to some polymers and just watched it take over the entire culture dish. It turned it all into these highly-organized liquid crystals.”
“These crystals have a quality that is almost infectious,” Bollyky said. When the crystalline bodies of these stringy phage come into contact with loose cellular debris, the debris falls into alignment with the liquid crystal.
James Wilking, a physicist who works on biofilm structure at Montana State University but who was not involved in this study, agreed about the need for bio-physics collaboration. “People really don’t understand the structure of the [biofilm] matrix or how to attack it,” he said. “[Physicist] people have known about the type of structures they saw for a while, but the fact that they saw it in a medically relevant biofilm is pretty cool.”
On the other hand, biologists point out that the notion of a phage supporting its bacteria is an important realization in its own right. “We tend to think of it as black and white or predators and prey, where the phage are obviously the prey,” said microbiologist Scott Rice from University of New South Wales. “I fully believe that evolution and selection will drive those organisms toward some sort of detente.”


