HIV proteins’ ‘social networks’ reveal why some patients control infection better than others
A new computational analysis identifies weak spots in the virus that causes AIDS, and could someday inform the development of a long-awaited vaccine.

Each protein can be seen as a "social network" of interconnected parts. The best bet for destroying a protein (and, perhaps, whatever that protein forms of the foundation of) is to hit the most "networked" pieces; if these break, the entire structure can unravel. Image Credit: gremlin, iStock
In 1994, physician-researcher Bruce Walker was sure he’d stumbled upon the impossible.
Despite being HIV positive, the patient in his office was healthy—and had been for at least a decade, all without the help of drugs. But in a year when AIDS would kill 35,000 people in the United States alone, all Walker’s patient wanted to know was when he would die, too.
Walker had no answer for him. It was the first time he had seen someone subvert what he’d been certain was a death sentence. “I was stunned,” Walker recalls. “I just thought, ‘This is unbelievable. This means HIV doesn’t kill everyone.’”
In the years that followed, Walker, now a researcher at the Ragon Institute, would come to realize that this remarkable patient wasn’t a fluke. Thousands more like him existed, many of whom were completely unaware they harbored the virus in the first place—all because their immune systems had somehow kept the deadly virus in check.
This singular riddle has driven Walker’s research for the past 25 years. But at long last, he and his team think they’re close to an answer. In a study published today in the journal Science, they propose an explanation for why, despite being exposed to the same virus, some individuals control infection (“controllers”), while others progress to disease (“progressors”). [Disclosure: Walker is also a relative of NOVA’s Deputy Executive Producer, Julia Cort.]
According to the study, hitting HIV’s Achilles heel may require leveraging the “social network” that governs the virus’ structure. HIV, like all viruses, is made up of several interconnected pieces. Much like the pieces in a chess game, those parts aren’t equally influential—which means an effective immune response is likely to be one that pinpoints the most pivotal players on the board.
“This is a very elegant piece of science that could identify potential weak spots in HIV,” says Nilu Goonetilleke, an immunologist studying HIV at the University of North Carolina’s School of Medicine who was not involved in the study. “I look forward to seeing a design for a vaccine come out of this, and, hopefully, seeing it tested.”
A scanning electromicrograph of an HIV-infected cell. Image Credit: NIAID, flickr
HIV is particularly tricky for the immune system to deal with. Not only does the virus infect and incapacitate some of the very immune cells that might otherwise defend against it, but it also behaves a bit like a moving target.
When on the lookout for pathogens like viruses, the immune system scours the body for foreign molecules with incriminating features—the microscopic equivalent of passing a mugshot from cell to cell. But when the HIV virus invades cells to copy its genome, it does so sloppily, accumulating so many mutations it can change the way it appears to the immune system. The HIV of one cell could look completely different from the HIV of another—and any intel the immune system may have stored can quickly become obsolete, allowing the virus to slip by unnoticed.
As it passes from person to person, the virus shapeshifts further, generating many distinct subgroups within and across populations. And protection against one version of HIV doesn’t guarantee protection against another.
In the face of such staggering diversity, an effective HIV vaccine will have to accomplish what the typical immune system cannot—recognizing the virus in all its myriad forms, says Morgane Rolland, an HIV virologist and vaccinologist at the U.S. Military HIV Research Program who was not involved in the study.
But even across vastly different strains, certain features of HIV remain the same, and don’t mutate as easily as others. Researchers think these so-called “conserved” spots represent indispensable components of HIV anatomy—ones that help it break its way into cells, or manufacture more of itself once inside—that could cripple the virus if altered.
Several years ago, Walker’s group found that these immutable regions seemed to be the same ones targeted by the immune systems of many HIV controllers. But Walker was disappointed to find the relationship was imperfect: Some of the patients who were homing in on seemingly crucial pieces of the virus were still getting sick.
So his team decided to go deeper, and zoom in on some of the proteins that contained conserved regions. They quickly discovered that, while all conserved regions were slow to change, only some of them had something else in common: They wielded an enormous amount of influence on the rest of the protein.
Whether they’re mingling with coworkers or counting degrees of separation from Kevin Bacon, every human is a member of a social network: a complex constellation of interpersonal relationships. For better or worse, social networks tend to distribute power unequally, with certain members who are better connected, more well liked, or who simply hold more sway over others.
The same, Walker says, is true of proteins like the ones that make up HIV. Though each protein is first synthesized as a linear chain of subunits called amino acids, it eventually takes the form of a three-dimensional structure, transforming what’s essentially a string of molecular “beads” into a web of convoluted loops and whorls. Amino acids that are nowhere near each other in the chain can come close enough to touch, creating an intricate nexus of associations that can bridge enormous divides. In other words, a network.
But clout can be a double-edged sword: In any network, the same pieces that hold it together are the ones it can least afford to lose. Walker’s team found that the more partners an amino acid had, the more central it was to a protein’s structural integrity—which meant snipping at just one of these critical threads could be enough to unravel the entire viral tapestry. Using a computer algorithm, the researchers analyzed the sequence of several HIV proteins and assigned each amino acid a “score” denoting how networked it was. When the team then mutated the amino acids with the highest scores, the entire virus “basically fell apart,” Walker says.
“Networked amino acids are the ones that are touching everything else,” says Mohammed AlQuraishi, a structural biologist at Harvard University who was not involved in the study. “If you want to target a large number of strains, these are your best bets.”
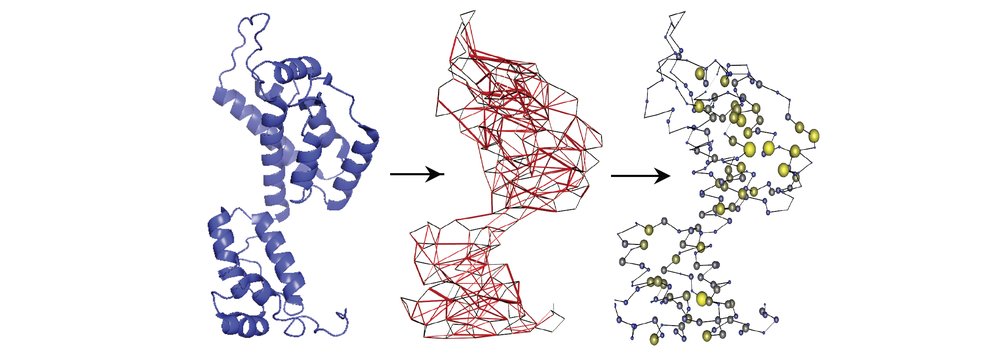
An HIV protein’s 3D structure (left) is analyzed to produce a map of the connections between amino acids (center). At right, the larger circular nodes represent the more connected amino acids. Image Credit: Adapted from Gaiha et al., Science, 2019
In a final test, the team discovered that the immune cells of HIV controllers, but not progressors, consistently zeroed in on highly networked amino acids. It seemed the team had finally found what distinguished the patients who could fight off infection: their ability to root out the Kevin Bacons of the HIV protein grid.
Walker’s team is now working to design a vaccine based on these same principles. The hope is to develop something that can train just about any immune system to single out these highly networked amino acids, says study author Gaurav Gaiha, a physician at Massachusetts General Hospital. This would saddle HIV with a serious dilemma: Mutate into impotence, or stay the same and be caught red handed.
That’s still a long way off. Vaccine design involves a lot more than earmarking an immunological bulls-eye, Rolland says. In the coming years, the team will have to show that they can actually redirect a person’s immune response without compromising safety. What’s more, the best vaccines will ideally rouse multiple branches of the immune system, including those that produce antibodies, which were not tested in this study.
Additionally, there are person-to-person differences to consider. “Even if two people target the exact same things, the quality of the immune response can still be very different,” Goonetilleke says.
For now, though, Walker is optimistic. Vaccine or not, the end of a 25-year odyssey could finally be in sight. “After years of searching, we’re finally able to understand this remarkable outcome in some people who are HIV infected,” he says. “I feel like I finally understand what was going on in that patient I first met in 1994.”