A second patient has entered long-term remission from HIV
Twelve years after the first man was declared to be rid of HIV, a second patient has achieved a similar milestone, sparking hope that a cure may someday be possible.
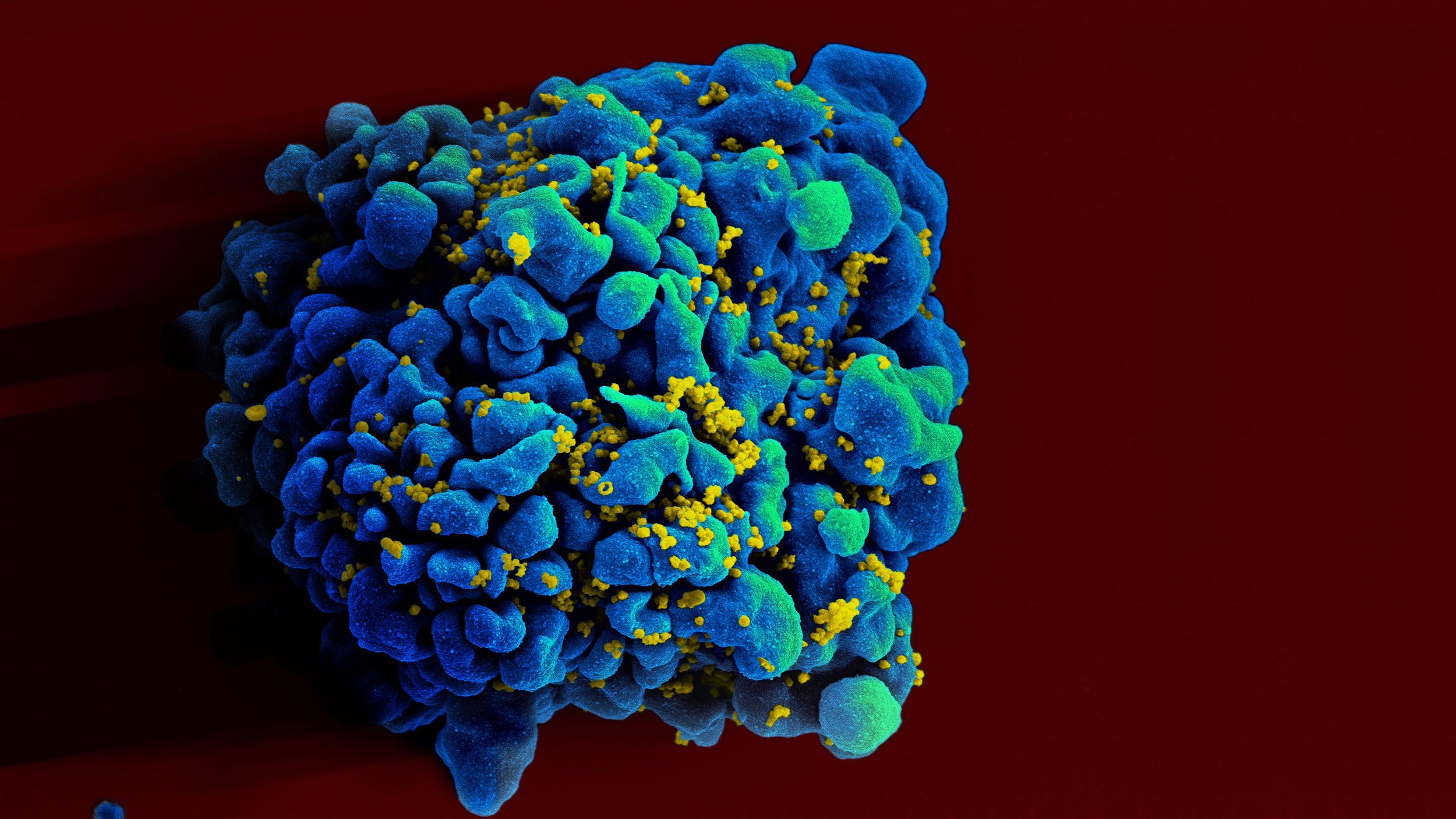
A T cell infected with HIV, the virus that causes AIDS. T cells play a critical role in the body's immune response, and when compromised by infection, can leave patients vulnerable to disease. Twice in history, stem cell transplants from donors with HIV-resistant cells have successfully put patients into long-term remission from the virus. Image Credit: NIAID, flickr
For the second time, a patient is in long-term remission from HIV, the virus that causes AIDS. With the help of a stem cell transplant from a virus-resistant donor, the anonymous individual—referred to by researchers as the “London patient”—has now been off antiretroviral drugs for about a year and a half.
The procedure is far from a panacea, however. This particular intervention is likely to be effective in only a small fraction of the 37 million people worldwide who currently harbor the virus in their bodies. Even so, the announcement, published today in the journal Nature, represents a long-awaited advance, in part by reaffirming that a similar case reported 12 years prior “wasn’t a one-off,” Sharon Lewin, who heads the Peter Doherty Institute for Infection and Immunity in Melbourne, Australia and was not involved in the study, told Jon Cohen at Science.
In several ways, the newly reported London patient echoes the first case of sustained remission from HIV: Timothy Brown, known in medical circles as the “Berlin patient,” who was effectively rid of the virus in 2007. Both milestones were achieved following a transplant of bone marrow, where the immune cells most susceptible to the virus originate, from a person with a mutation in the gene that encodes CCR5, a protein that many HIV strains use to infiltrate cells. In other words, the procedure effectively swapped out vulnerable tissue for hardier stock. Notably, neither treatment was originally intended to eliminate the infection itself, but to treat blood cancers that had spread in both individuals.
A few key differences exist between Brown’s case and that of the London patient, however. To complete the transplant, doctors administered harsh immunosuppressive drugs, causing him to suffer severe complications in the months after receiving the cells. At one point, Brown had to be placed in an induced coma and nearly died.
The London patient, on the other hand, was able to undergo a far gentler regimen with similar results, though the broad therapeutic strokes were more or less equivalent. This crystallizes the idea that invasive, life-threatening treatments aren’t required to achieve remission from HIV.
“I think this does change the game a little bit,” study author Ravindra Gupta, a virologist at University College London told Apoorva Mandavilli at The New York Times. “Everybody believed after the Berlin patient that you needed to nearly die basically to cure H.I.V. But now maybe you don’t.”
On the whole, though, bone marrow transplants remain risky and expensive, and, apart from the two aforementioned patients, have a poor track record of clearing HIV from the system, despite numerous attempts. As such, this procedure is unlikely to become a realistic treatment for HIV in the future—especially given that the antiretroviral drugs prescribed for the infection have been shown to be extremely effective. “[This] doesn’t change things for the average person with HIV right now,” the Ragon Institute’s Bruce Walker, who was not involved in the study, told Kate Sheridan at STAT News.
However, current treatments must be administered throughout a patient’s life, and millions worldwide lack access to a source of consistent therapy. On top of this, drug-resistant strains of HIV continue to surface, throwing a wrench in current formulations.
As such, the findings provide a glimmer of hope for researchers who plan to build on this knowledge to explore new treatment options. One possibility would be to alter the CCR5 gene, and thus compromise one of HIV’s most prominent entry points into human cells. This is not a new idea: the CCR5 gene was infamously targeted by Chinese scientist He Jiankui, whose (possibly failed) attempt to edit human embryos to make them immune to HIV sparked an international scandal last fall.
He’s experiment was widely condemned as unethical and reckless. But many other groups of researchers are taking more cautious approaches to manipulating CCR5.
Still, CCR5 is not the only gateway for HIV: Some strains are able to bypass this molecule entirely on their way into cells. But if achievable, such treatments could set an important precedent going forward.

Though modern antiretroviral drugs are effective, prescribed regimens are lifelong, and not all communities worldwide have access to treatment. Image Credit: NIAID, flickr
Though the London patient is choosing not to disclose his name for the time being, he described the experience as “surreal” and “overwhelming” in an email to Mandavilli at The New York Times. “I never thought that there would be a cure during my lifetime,” he wrote.
Of course, remission is not necessarily a “cure” (though many have referred to this landmark achievement as such), Gupta told Cohen at Science. The researchers drew their conclusions based exclusively on the London patient’s blood, and will continue to monitor his health in the years to come.
In the meantime, though, the team is tentatively optimistic. And Timothy Brown, who has held a solo spotlight for the last 12 years, is relieved to finally have someone to share it. “If something has happened once in medical science, it can happen again,” Brown told Mandavilli at The New York Times. “I’ve been waiting for company for a long time.”



