To save a girl’s life, researchers injected her with genetically engineered viruses
So-called phage therapy isn’t yet mainstream, but in some cases it might be the only option for antibiotic-resistant bacterial infections.

Bacteriophages, or phages for short, are viruses that exclusively infect bacteria. Because they're extremely picky about their hosts, phages could have potential as a very precise medical treatment for antibiotic-resistant bacteria infections. Image Credit: Shutterstock
Even before Rebekah Dedrick unpacked the samples, the countdown had already begun.
Some 4,000 miles from her lab in Pittsburgh, at the Great Ormond Street Hospital in London, a 15-year-old girl with cystic fibrosis was battling a life-threatening infection. After receiving a double lung transplant, the young patient had been put on a regimen of immunosuppressive drugs, and an obstinate bacterium called Mycobacterium abscessus had taken hold. The microbes proved resistant to every antibiotic the girl’s doctors tried, and were now spreading throughout her body.
In a last-ditch effort, the London medical team shipped tubes teeming with bacteria isolated from the patient to Dedrick and her colleagues, microbiologists at the University of Pittsburgh. The samples came with an urgent—and radical—request: Identify a swarm of viruses that could be injected into the girl’s body to kill the drug-resistant bacteria.
It was risky move. This virus-based technique, called phage therapy, had never been used against the genus Mycobacterium—a group that includes the bacteria that cause tuberculosis and leprosy—making the method entirely experimental. But as Dedrick and her colleagues report today in the journal Nature Medicine, their gamble seems to have paid off: A year after the first-ever use of genetically engineered phages against a Mycobacterium microbe in the clinic, the patient is alive and well.
So far, the procedure has only been conducted on a single patient, and will require vetting and refinement through clinical trials before it has the chance to enter mainstream medical use. With these caveats in mind, however, “this study is foundational for the future of this field,” says Marisa Pedulla, a phage researcher at Montana Tech who was not involved in the study. “All future studies with mycobacteria and phages will cite this key work.”
Though it remains controversial in most parts of the world, phage therapy isn’t a new idea. Since the early 1900s, scientists have been aware that microbes have their own assailants, in the form of viruses that hijack the molecular machinery inside bacterial cells for their own reproductive purposes. Because they invade and destroy their hosts, these “bacteria-eating” viruses—bacteriophages, or phages for short—can be cataclysmic forces of change for microbes, reshaping entire populations through periodic massacre.
Phages, however, are also notoriously picky: A strain that infect one species (or even subspecies) will often snub another type of bacterium that deviates even slightly from their preferred host. Combined with these viruses’ lethal modus operandi, this precision is exactly what gives phages such potential as a treatment. Unlike antibiotics, which operate fairly indiscriminately, phages could, in theory, annihilate the sole culprit behind an infection while leaving everything else intact; then, with their targets depleted, phages would be hastily cleared from the body.
Though the practice remains controversial and mostly experimental, phage therapy hasn’t been without its victories: In 2016, experimental phage therapy was successfully used as an emergency treatment for a drug-resistant Acinetobacter baumannii infection.
Phages carry genetic material, but can't replicate on their own. To make more of themselves, they infect bacterial cells, often killing them in the process. Image Credit: Shutterstock
This was the hope that motivated the London medical team when they first approached Dedrick and her advisor, Graham Hatfull, in the fall of 2017. Hatfull’s lab at the University of Pittsburgh had spent the past decade stockpiling a repository of more than 10,000 different types of phage active against mycobacteria, most collected over the years by high school and undergraduate students in the Science Education Alliance Phage Hunters Advancing Genomics and Evolutionary Science (SEA-PHAGES) program. If the phage the doctors needed existed, their best chances of finding it were here.
There was just one catch. While all of Hatfull’s banked phages were known to infect a non-infectious cousin of Mycobacterium abscessus, none had actually been tested on M. abscessus itself. Given just how finicky phages can be, this left Dedrick and her colleagues with a lot of unknown candidates.
Undaunted, the team broke the phages down into dozens of genetically distinct groups and began to test representatives one by one. For weeks on end, the researchers worked day and night, smearing phage after phage onto opaque films of M. abscessus. All the while, the clock in London continued to tick. “It was a blur,” Dedrick recalls. “We were working as hard and fast as we could.”
Each morning, Dedrick and her colleagues scoured the plates for the telltale translucent spots where the viruses had successfully massacred the microbes below—but over and over, the bacteria continued to grow.
Until one day, they didn’t. In January of 2018, Dedrick was astounded to find a phage named Muddy had produced a small but unmistakable clearing in the otherwise thick lawn of M. abscessus. “When we saw Muddy for the first time, we were like, ‘Oh my gosh, we actually found something that infects this,’” Dedrick says. “We’d had so many let downs before that.”
But just as bacteria can evolve resistance to antibiotics, so, too, can they become immune to their viral assailants—and using just one phage would be too risky for such a dire case. In the months that followed, the researchers went on to identify two more phages, ZoeJ and BPs, that, with some genetic tinkering, also infected the patient’s M. abscessus strain. The three active phages were then purified, slurried into a cocktail, and shipped overseas.
In the meantime, however, several months had passed, and the patient had worsened. After multiplying in her newly transplanted lungs, the bacteria had spread to infect the surgical wound site, her liver, and more than 20 other locations on her skin. None of the dozens of antibiotics administered had made much of a difference, and her chances of survival dwindled to 1 percent.
“This was clearly a very serious case,” says Stacey Martiniano, a clinician at the University of Colorado School of Medicine who specializes in pediatric pulmonology, and was not involved in the study. “Not a lot of cystic fibrosis patients get this sick with M. abscessus...but all of us in research, as well as caregivers, have seen patients in these situations where we’re seeing the limitations of our antibiotics. It’s what led [these doctors] to try whatever they possibly could.”
With all other options exhausted and no precedent to guide them, the medical team in London deployed their final weapon. A small dose of the viral concoction was applied as a salve to infected areas on the patient’s skin, while the rest was injected with an IV in an attempt to drive the bacteria from her organs.
Nine days later, the patient was discharged from the hospital.
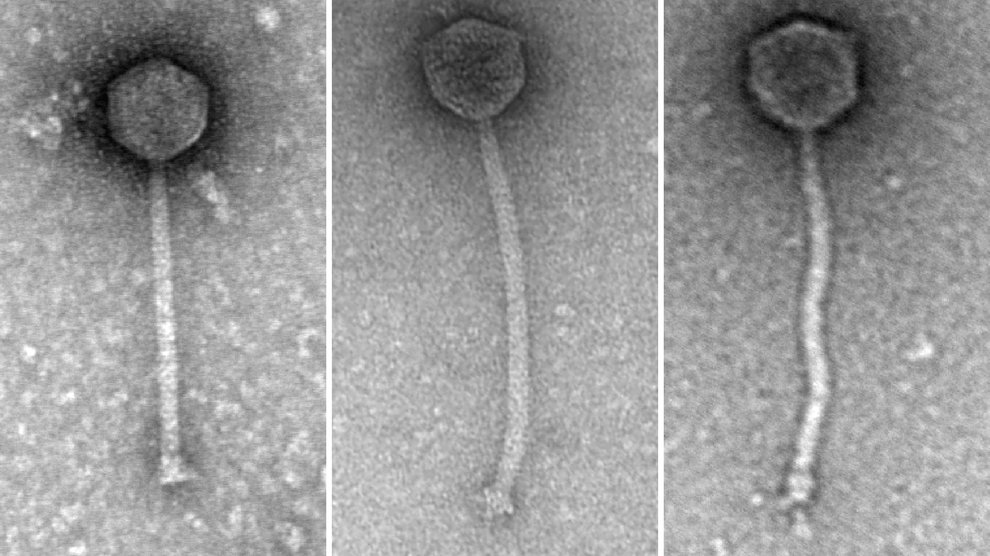
The Muddy, BPs, and ZoeJ phages, respectively, which were successfully used in combination as phage therapy for a 15-year-old patient in London in 2018. Image Credit: Courtesy of Rebekah Dedrick, Graham Hatfull Lab, University of Pittsburgh
That was a year ago. In the months since, with continued phage therapy, the patient’s wounds and infections have healed, her weight has gone up, and she’s recovered function in her liver and lungs. The phages, it seems, did their job.
“This illustrates something very important—that it is possible to administer phage safely and have a good clinical outcome,” Hatfull says. “That hasn’t been shown before for these types of infections. That means there’s hope...in a sense, we’ve demonstrated that this can work.”
It wasn’t a cure, though—not exactly. While much of the M. abscessus was cleared from the patient’s system, her phage therapy is ongoing, Dedrick says. It’s not yet clear when enough will be enough.
And with just one patient in the pipeline, there’s no telling what the future of this treatment will be, or even if it will be effective or safe for other individuals. “Case studies are really important and exciting—they’re a way to bring to light rare treatments and cases and bring researchers together,” Martiniano says. “But we need other research strategies, like clinical trials.”
Those are hopefully to come, Hatfull says. One challenge is that this particular treatment is not generalizable—even among M. abscessus infections. The same precision that makes phage therapy such a powerful tool might also be its greatest weakness: A given virus might be useful against only a small number of bacterial strains. This stringency could necessitate rejiggering the viral cocktail for each individual patient, as well as costly maintenance of phage banks worldwide to meet changing demands.
While this “personalized medicine” approach has its benefits, many patients won’t be able to afford the time it takes to sift through a vast library of phages, isolate viruses, and ready them for treatment, says Chidiebere Akusobi, an infectious disease researcher studying M. abscessus at Harvard University who was not involved in the study. “Individualized treatment takes time to individualize,” Akusobi says. “The patient was probably lucky to still be alive while her cocktail was being generated.”
The downside of timing isn’t just a hypothetical. Over the course of the study, the researchers encountered another patient carrying a genetically distinct strain of M. abscessus. But none of the phages they tested could hack it against the bacteria, and the second patient died.
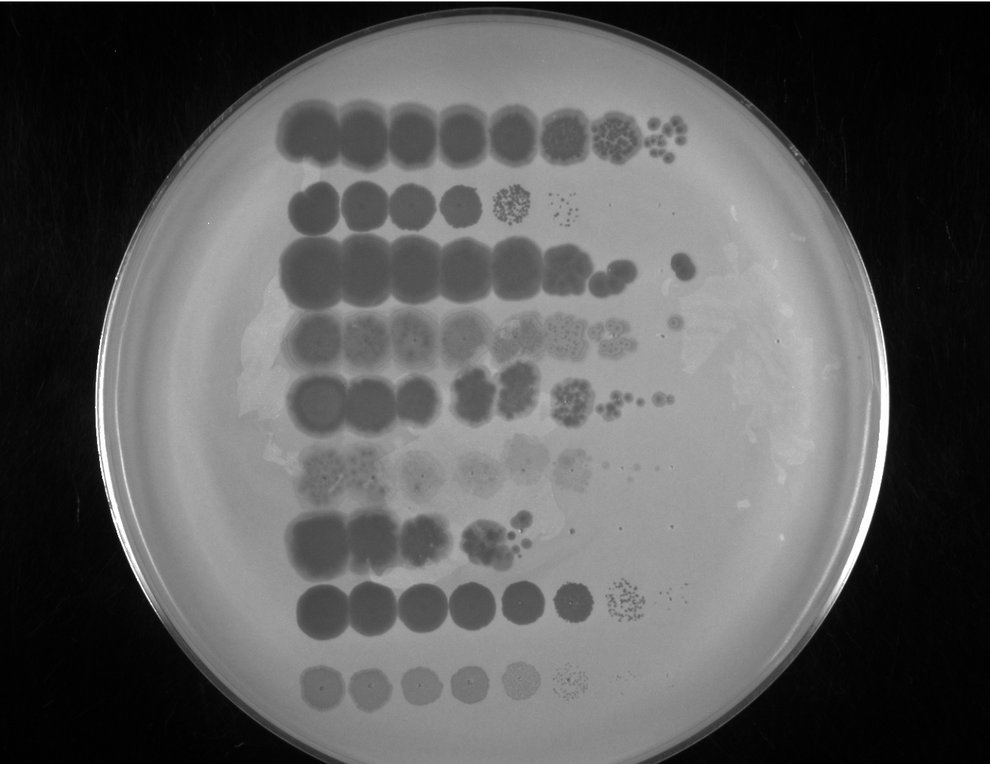
An agar plate coated with Mycobacterium smegmatis, a cousin of Mycobacterium abscessus. Each row of spots shows a different type of phage, becoming more diluted from left to right. Clear spots show where the phage killed the bacteria below. Image Credit: Courtesy of Rebekah Dedrick, Graham Hatfull Lab, University of Pittsburgh
Even if they’re identified in time to treat a patient, phages aren’t foolproof. Bacteria are a constantly moving target, and can still evolve resistance against phages (and pretty much any other treatment, for that matter). And though they mean no harm to human cells, phages—like any other virus—can still invoke the wrath of the immune system. Because this patient didn’t seem to have serious side effects, it doesn’t seem like that was the case here, Akusobi says, but it should come as no surprise that “injecting large amounts of foreign particles into a body can be dangerous for some.”
Implementing phage therapy on a larger scale will also require closing huge gaps in knowledge. Phages vastly outnumber any other biological entity on Earth (estimates of their numbers worldwide run around 10 nonillion—that’s 1 with 31 zeros), and scientists don’t yet have a sense for the diversity that’s out there. Even the few phages that researchers have banked remain mostly enigmatic, says Asma Hatoum-Aslan, a phage researcher at the University of Alabama who was not involved in the study. This uncertainty makes every injection of phages into a human body a near-literal shot in the dark.
Regardless, the new study is “very exciting and important,” says Hatoum-Aslan, who remains optimistic about the future of phage therapy. Though the procedure in the study will require further refinement, this early success hints that methods like these could someday enhance, complement, or follow antibiotic treatment for these microbes and more.
And for now, even a single victory is still, well, a victory—especially for the patient. “Just seeing her before treatment, and now after...I could see the improvement with my own eyes,” Dedrick says. “That just makes me really proud.”



