|
|

|
 |
Here's what happens when you mix two of the materials, oil and water, together: (you'll see the action four times) 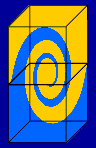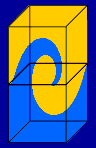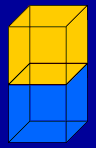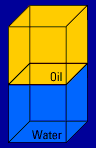 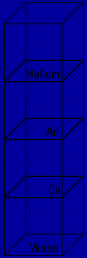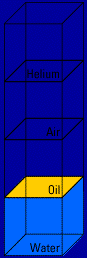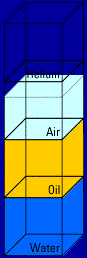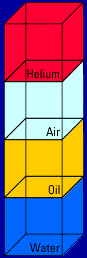 Look what floats and what sinks! Helium floats on top of
everything because it has the lowest density. And the water
sinks to the bottom because it has the highest density.
Look what floats and what sinks! Helium floats on top of
everything because it has the lowest density. And the water
sinks to the bottom because it has the highest density.Think about these everyday demonstrations of density . . . A birthday balloon full of helium: Helium has a lower density than air. That's why a helium balloon rises in air. A scuba diver under water: Air has a lower density than water. That's why the diver's air bubbles rise to the surface of the water. A puddle with an oil slick on top: Did you ever notice how streaks of oil sit on top of a puddle? That's because the oil has a lower density than water, so it rises to the top of the puddle. "But what about hot air?" you're probably saying. Well, you already know that a hot air balloon floats. What, then, would you conclude?
Global Contenders '97/'98 | Expedition '96/'97 | Fossett | Virtual Flight Science of Ballooning | Teacher's Guide | Resources | Transcript | Balloon Home Editor's Picks | Previous Sites | Join Us/E-mail | TV/Web Schedule About NOVA | Teachers | Site Map | Shop | Jobs | Search | To print PBS Online | NOVA Online | WGBH © | Updated October 2000 |