How a Drug Company Pushed Opioids & Made Millions by Bribing Doctors & Committing Insurance Fraud
June 19, 2020
Share
In a new documentary and related reporting, FRONTLINE and the Financial Times investigate the spectacular rise and fall of Insys Therapeutics
Opioids, Inc.
Streaming online beginning Fri., June 19, 2020
Premiering on PBS stations Tues., June 23, 2020
www.facebook.com/frontline | Twitter: @frontlinepbs
Instagram: @frontlinepbs | YouTube: youtube.com/frontline
What role have pharmaceutical companies played in fueling America’s epidemic of opioid addiction, and how have they and their stockholders profited?
FRONTLINE and the Financial Times (FT) present Opioids, Inc., a documentary revealing how one drug company bribed doctors, committed insurance fraud and made millions for Wall Street investors pushing a highly addictive opioid painkiller — and how it then became the first pharmaceutical company to have its CEO sentenced to prison time in federal court in connection with the opioid crisis.
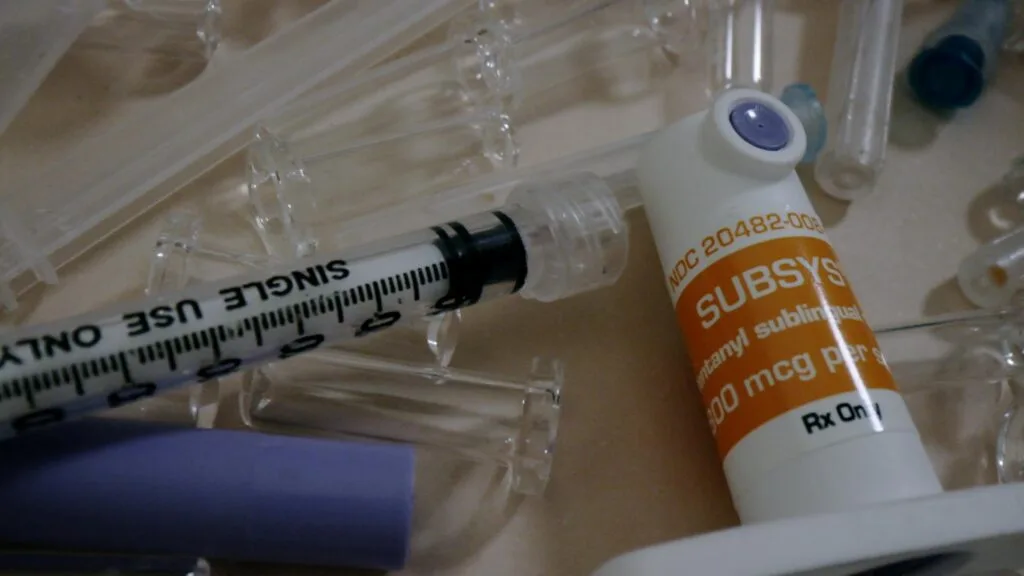
The company was Insys Therapeutics, the CEO was John Kapoor, and the drug was Subsys, a fast-acting fentanyl-based spray that is 50 to 100 times stronger than morphine. Approved for treating cancer pain, it was prescribed much more generally, helping the company’s sales reach more than $300 million at their peak and stock prices on Wall Street surging.
“When a company’s share price goes ballistic like that, yes it may be because they discovered a new innovation that is so exciting and good for mankind that it’s going to carry on indefinitely, but that’s a pretty rare example,” FT editor at large Gillian Tett says in Opioids, Inc. “Usually, there is something in that, that’s just too good to be true.”
This yearlong investigation, which also includes an in-depth story from the FT publishing June 19, is the first collaboration between FRONTLINE and the FT. The Opioids, Inc. documentary will be available to stream beginning June 19, and will premiere on PBS stations June 23 (check local listings).
In the documentary, with firsthand accounts from key players, award-winning FRONTLINE director Tom Jennings and his reporting and producing partners tell the inside story of the corruption behind Insys’ spectacular rise — a scheme that federal prosecutors said went all the way to the top, and that involved paying doctors to prescribe extreme doses of Subsys to their patients — and how investors looked the other way.
“There were people on Wall Street who were intent on keeping this stock price up, and I believe ignoring some of the more obvious indications that this was an unsustainable, potentially fraudulent business model,” says Jim Carruthers, one of a small number of investors known as short sellers who were skeptical of Insys.
In unprecedented detail, Opioids Inc. goes inside that business model: “The instruction was, go out there, show that we can get a minimum return on investment of 2:1, minimum, and do not lose [Kapoor’s] money or get fired. And the only way that I knew how to do it to get that guarantee, is to bribe doctors,” Insys’ former director of sales Alec Burlakoff tells FRONTLINE and the FT in his first on-camera interview.
Burlakoff is one of eight people connected to Insys and Kapoor to speak out in the film with their experiences.
“I believed that he was somebody that was able to help us change the way we have cancer pain treatment in America,” pain management specialist Dr. Lisa Jo Stearns, who ran one of the first clinical trials of Subsys, says of her initial impression of Kapoor. She recalls him tearing up as he discussed the pain experienced by his wife as she died of cancer.
But former sales representatives from Kapoor’s company describe a culture of unbridled greed, detailing to FRONTLINE and the FT how they targeted high-prescribing doctors and nurse practitioners known as “whales.”
“It wasn’t about cancer patients. It was about getting as many people as you could on the drug,” says former sales representative April Moore, adding, “Low doses aren’t that much money. Higher dose, more money.”
The company even held contests for the sales team: the higher the doses they got doctors to write, the larger the cash prize — despite the dangers to patients.
“This is probably the most guilty thing in the company. If you had a doctor who wrote a prescription, it was a bonus on your paycheck. If you reach the 1600 mcg of fentanyl, that could kill you, but people were getting there,” says another former sales representative, Tim Neely.
At the same time, the company was misleading insurers to approve prescriptions of the drug: “None of what we were saying was truthful,” a former prior authorization specialist says. “We’re just pocketing the money off of a prescription that should’ve never been approved anyway. That’s insurance fraud.”
Opioids, Inc. examines how federal prosecutors prepared the case against Insys by pursuing a novel strategy, using anti-racketeering laws designed to fight organized crime and working their way up the company’s ranks — and how they ultimately arrived at a “smoking gun”: a spreadsheet ordered by Kapoor that showed how Insys tracked the money that went to doctors, and what the company got in return.
“I called it a smoking gun. I mean, it was about this return on investment theory that Kapoor had, and that’s criminal. So you might as well be writing out a confession,” says federal prosecutor Fred Wyshak, who had previously been renowned for prosecuting mob kingpins like Whitey Bulger.
With prison terms handed down for Kapoor, Burlakoff and other former Insys executives, other drug makers and distributors are now on notice as the opioid crisis continues.
Opioids, Inc. will be available to watch in full at pbs.org/frontline, on YouTube, and in the PBS Video App starting Friday, June 19. An in-depth Financial Times story will publish the same day at FT.com and at pbs.org/frontline. The documentary will premiere on PBS stations on Tuesday, June 23 at 10/9c (check local listings).
###
Credits
Opioids, Inc. is a FRONTLINE production with 2Over10 Media in association with the Financial Times. The writer and director is Thomas Jennings. The producers are Annie Wong and Nick Verbitsky. The journalist from the Financial Times is Hannah Kuchler, with reporting credit to US Editor of Alpha Grid Shaunagh Connaire. The co-producers are Anna Auster and Shaunagh Connaire. The senior producer is Frank Koughan. The executive producer of FRONTLINE is Raney Aronson-Rath.
About FRONTLINE
FRONTLINE, U.S. television’s longest running investigative documentary series, explores the issues of our times through powerful storytelling. FRONTLINE has won every major journalism and broadcasting award, including 93 Emmy Awards and 24 Peabody Awards. Visit pbs.org/frontline and follow us on Twitter, Facebook, Instagram, and YouTube to learn more. FRONTLINE is produced by WGBH Boston and is broadcast nationwide on PBS. Funding for FRONTLINE is provided through the support of PBS viewers and by the Corporation for Public Broadcasting. Major funding for FRONTLINE is provided by The John D. and Catherine T. MacArthur Foundation and the Ford Foundation. Additional funding is provided by the Abrams Foundation, the Park Foundation, the John and Helen Glessner Family Trust and the FRONTLINE Journalism Fund with major support from Jon and Jo Ann Hagler on behalf of the Jon L. Hagler Foundation.
About the Financial Times
The Financial Times is one of the world’s leading business news organizations, recognized internationally for its authority, integrity and accuracy. The FT has a record paying readership of more than one million, three-quarters of which are digital subscriptions. It is part of Nikkei Inc., which provides a broad range of information, news and services for the global business community.
For more information, please contact:
FRONTLINE: Anne Husted / anne_husted@wgbh.org Financial Times: Katrina Fedczuk / katrina.fedczuk@ft.com
Latest Documentaries
Explore
Policies
Teacher Center
Funding for FRONTLINE is provided through the support of PBS viewers and by the Corporation for Public Broadcasting, with major support from Ford Foundation, and The Fialkow Family Foundation. Additional funding is provided the Abrams Foundation, Park Foundation, John D. and Catherine T. MacArthur Foundation, Heising-Simons Foundation, and the FRONTLINE Trust, with major support from Jon and Jo Ann Hagler on behalf of the Jon L. Hagler Foundation, and Corey David Sauer, and additional support from Koo and Patricia Yuen. FRONTLINE is a registered trademark of WGBH Educational Foundation. Web Site Copyright ©1995-2025 WGBH Educational Foundation. PBS is a 501(c)(3) not-for-profit organization.