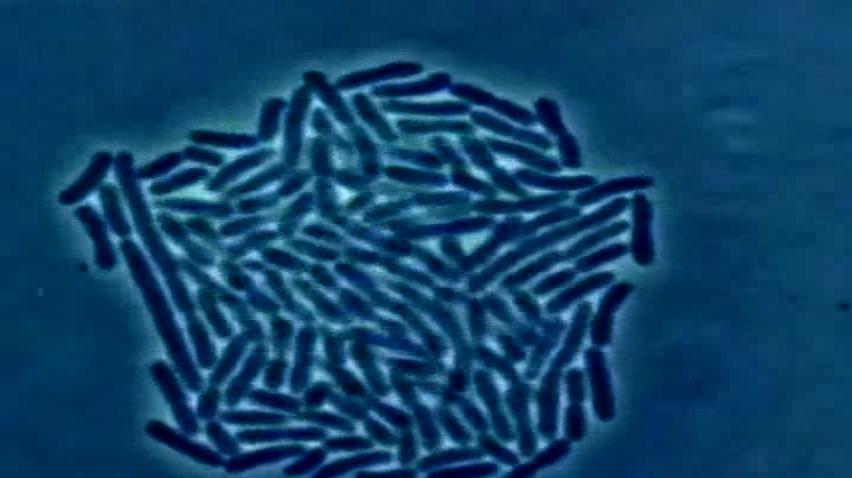
Lawmakers Take Aim at Link Between Antibiotics for Livestock and Infections in Humans

March 3, 2015
Share
The amount of antibiotics being sold to treat the nation’s livestock is on the rise, but what’s difficult to tell is just how much those drugs are fueling the spread of potentially deadly drug-resistant superbugs. In a bid to address that risk, three U.S. senators have introduced legislation that would expand the authority of the Food and Drug Administration (FDA) to pull such antibiotics from use.
The legislation, introduced Monday, would require the FDA to withdraw approval of any antibiotic used for the purpose of disease control or prevention in food-producing livestock, unless the drug’s manufacturer can show that its use in animals won’t harm human health. The bill is designed to close a gap in a set of guidelines issued by the FDA in 2013 calling on drug makers to voluntarily end the use of antibiotics for the goal of helping animals to gain weight — a strategy often used to raise animals on less feed.
The bill would also establish limits on how long medications that maintain their FDA approval can be used for preventing or controlling disease. The FDA estimates that 107 antibiotics either have no limit on how long they can be used for or are labeled for continuous use. The fear among many scientists is that the longer an animal remains under a course of treatment that is no longer necessary, the more likely they may be to spawn a drug-resistant bacteria of some sort.
“Antibiotic resistance is one of the biggest public health threats we face and we need a comprehensive response to preserve the effectiveness of antibiotics,” said Sen. Dianne Feinstein (D-Calif.), one of the measure’s three sponsors. “While FDA took an important step to reduce antibiotics overuse in agriculture, we need to do more.”
The bill, which was cosponsored by Senators Susan Collins (R-Maine), Kirsten Gillibrand (D-N.Y.) and Elizabeth Warren (D-Mass.), comes at a time when data from the Centers for Disease Control and Prevention finds that more than 20,000 people every year die due to an antibiotic resistant infection, and 2 million get sick.
The bill’s prospects remain far from certain, however. Legislation to address the use of antibiotics in livestock has been introduced at least six times since 2003, but the only measure to survive was a 2008 bill requiring the FDA to report sales figures on antibiotics used in animals.
In October, the agency announced that sales of antibiotics for agriculture climbed 16 percent between 2009 and 2012, meaning that up to 70 percent of all antibiotics now sold in the U.S. are used in food animals. Yet even though the agency has a better grasp of how many antibiotics are being sold, the string of legislative defeats has left the agency unable to answer with any specificity basic questions such as which drugs are being used, for what purpose, in which animals, and at what doses.
The early response from drug makers suggests the bill may face an uphill climb. In a statement to The Wall Street Journal, the Animal Health Institute, which lobbies on behalf of companies that produce medications for livestock, said:
Our members and many other stakeholders are engaged in working with FDA on successful implementation of the agency’s judicious use policy … We believe that is a more efficient way to achieve the apparent broad goals of this legislation.
Related Film: The Trouble With Antibiotics
FRONTLINE investigates the growing crisis of antibiotic resistance.

Related Documentaries
Latest Documentaries
Related Stories
Related Stories
Explore
Policies
Teacher Center
Funding for FRONTLINE is provided through the support of PBS viewers and by the Corporation for Public Broadcasting, with major support from Ford Foundation, and The Fialkow Family Foundation, as part of the Plum Bush Foundation. Additional funding is provided the Abrams Foundation, Park Foundation, John D. and Catherine T. MacArthur Foundation, Heising-Simons Foundation, and the FRONTLINE Trust, with major support from Jon and Jo Ann Hagler on behalf of the Jon L. Hagler Foundation, and Corey David Sauer, and additional support from Koo and Patricia Yuen. FRONTLINE is a registered trademark of WGBH Educational Foundation. Web Site Copyright ©1995-2025 WGBH Educational Foundation. PBS is a 501(c)(3) not-for-profit organization.