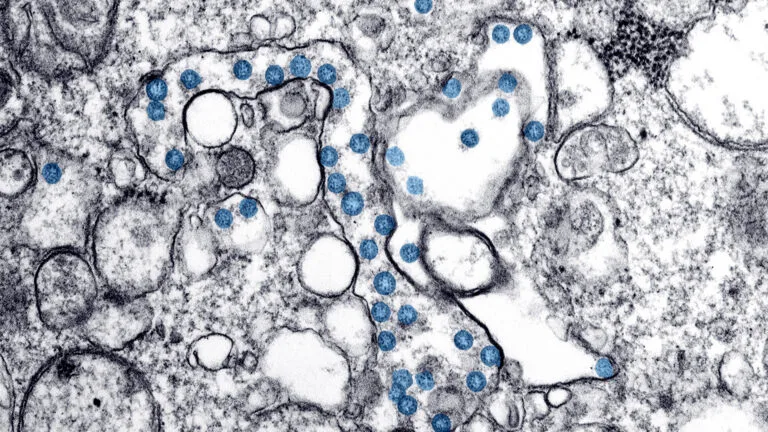
“This Is the New Reality”: Reporting on the Coronavirus from Washington State

March 19, 2020
Share
Miles O’Brien is reporting for the upcoming FRONTLINE, Coronavirus Pandemic, which is scheduled to air April 21 on most PBS stations at 9 p.m. EST/8 p.m. CST. Check local listings.
Day one covering the coronavirus: I’m awakened at 5:30 a.m. in Seattle by a call from the east coast from a person who didn’t know I was three hours behind. When I try to say hello, nothing comes out. My voice is gone. Normally this wouldn’t worry me, but given the context of the times and my location on the planet, I quickly move into high alert.
I take my pulse (normal), my temperature (normal), scan the web (as I’ve done so many times preparing for my trip here), and check the key symptoms of COVID-19.
This is the new reality for us all: the fear and hyper-alertness to signs of the coronavirus. I have none of those signs, and before long, the sun is rising, my voice is back, and I’m out the door to start my reporting for the film I’m working on for FRONTLINE.
I’ve got a small, dedicated crew with me here, who all decided to risk leaving their homes and families to tell this important story from the epicenter of the outbreak in the U.S.
Our first stop is north of Seattle, at the Providence Regional Medical Center in Everett, where the novel coronavirus is believed to have first come to U.S. soil in January. The 35-year-old man who fell ill had spent six weeks in the Wuhan region of China and developed symptoms a day after he returned from his trip. This is where he was treated.

Outside the hospital, we were immediately struck by what we saw: cars in line for drive-up coronavirus testing. They were employees of the hospital system who are, of course, particularly vulnerable to the spread of the virus.
When I left Boston a day earlier, these drive-up testing sites had yet to appear on the east coast; now they are springing up around the country as the virus takes hold.

We are adhering to a strict protocol while reporting: lots of hand sanitizer, often wearing masks, keeping a safe distance from interviewees, and generally treading carefully as we go. I watched as a woman named Kelsey came for testing. She normally works at the front desk of the hospital, but symptoms of COVID-19 have forced her into quarantine at home. A nasal swab is not a lot of fun, and Kelsey didn’t hide her displeasure. But by the time I spoke to her, her spirits were surprisingly good, even though she is a little bit scared.
“It’s a little daunting,” she told me. “But if you feel sick and you stay home, then you’re protecting yourself from getting something worse, but you’re also protecting other people. So, I mean, people just have to take the right steps.”
Inside, we met with Darren Redick, the executive director for operations. He walked us into the command center, where decisions are made about matching beds and patients. Right now, this 600-bed facility is at about 90 percent capacity. That’s actually a typical number. But what isn’t typical is that elective procedures have been canceled, which normally represents a significant portion of the inpatient bed count.
One of the floors usually dedicated to people recovering from surgery is now solely assigned to patients with COVID-19. It turns out that when the hospital was built, it was designed to be re-purposed for a health crisis like a pandemic. As a result, the patient rooms are all kept under negative pressure. This means the pressure in the hallways is higher, so whenever a door to a patient room is opened, the air rushes in, not out, as a way of containing the contagion.
It was on this COVID-dedicated floor that we met Dr. George Diaz, who treated patient number one.
He told me that in the weeks before the patient arrived, they had been simulating what they would do when — not if — the virus arrived at their hospital. So when it did, he and his colleagues sprang into action. That preparation has helped them with what has happened since. And it has put Diaz at the forefront of finding a way to emerge from the national nightmare.
To treat their first COVID-19 patient, Diaz took a risk and turned to an antiviral drug called remdesivir. It had proven safe for humans, but not necessarily effective at fighting viruses. Attempts to use it to treat Ebola, for example, were not successful. But as his COVID-19 patient got steadily worse and developed pneumonia, Diaz petitioned the FDA to allow the use of this drug under compassionate grounds. Within 24 hours, patient number one was receiving remdesivir intravenously. And only 24 hours later, his symptoms dramatically abated. He was released to home quarantine only a few days later.
Of course, this is a data point of one, an anecdote. No one is suggesting the drug will slow the spread of coronavirus. There are many, many questions. However, it was enough to spark a lot of interest in whether this drug might save lives in this pandemic. Diaz is now part of a multi-front urgent study underway to try to reach some scientifically valid conclusions. And the company that makes it is exploring ways to rapidly ramp up production.
Diaz likens this moment we are in now to the U.S. experience in World War II, when the country mobilized behind the war effort. He believes it’s time to take a wartime approach against coronavirus.
“If you look at, for example what happened with World War II, we’re bombed and immediately, the U.S. government basically converted all of our manufacturing into machines of creating the supplies necessary to fight a war,” he told me. “We have a lot of advanced research within the Seattle area. Those labs could have been potentially changed into labs to test for coronavirus on an industrial scale.”

There are a lot of unknowns in the days ahead — for the story, and for me and the team.
At Providence hospital, we were asked if we had a fever, a cough, generally felt poorly, and if we had contact with anyone diagnosed with COVID-19. They asked about recent international travel. They also took my temperature — 97.2.
Given how my day began, it was nice to keep hearing that number.
Related Documentaries
Latest Documentaries
Related Stories
Related Stories
Explore
Policies
Teacher Center
Funding for FRONTLINE is provided through the support of PBS viewers and by the Corporation for Public Broadcasting, with major support from Ford Foundation, and The Fialkow Family Foundation. Additional funding is provided the Abrams Foundation, Park Foundation, John D. and Catherine T. MacArthur Foundation, Heising-Simons Foundation, and the FRONTLINE Trust, with major support from Jon and Jo Ann Hagler on behalf of the Jon L. Hagler Foundation, and Corey David Sauer, and additional support from Koo and Patricia Yuen. FRONTLINE is a registered trademark of WGBH Educational Foundation. Web Site Copyright ©1995-2025 WGBH Educational Foundation. PBS is a 501(c)(3) not-for-profit organization.