What is the FDA Doing About the Problem?

October 14, 2014
Share
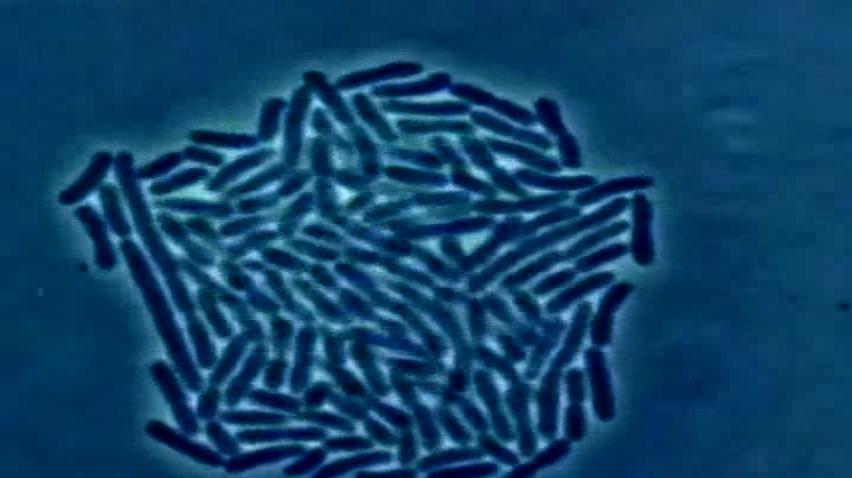
For nearly 40 years, public health officials and scientists have been pushing the Food and Drug Administration (FDA) to limit the use of antibiotics fed to animals raised for meat, a practice many worry is contributing to the rise of potentially deadly, drug-resistant infections.
Now, the agency is trying a different approach: asking companies to do so voluntarily.
The new policy comes at a time when the global health community has watched with alarm as many common antibiotics developed to fend off human illness have been rendered either ineffective or obsolete due to overuse. The Centers for Disease Control and Prevention estimates that more than 20,000 people die each year due to antibiotic-resistant infections, and as many as 2 million get sick. The FDA’s worry is that antibiotics used on the farm may be partly to blame.
Most antibiotics aren’t used by humans. Data compiled by the Pew Charitable Trusts, which advocates to limit antibiotic use on farms, shows that in 2011, 7.7 million pounds of antibiotics were sold to treat sick people, while a whopping 29.9 million pounds were sold for use in food animal production. In 2012, that figure grew to 32 million pounds, according to a report released earlier this month by the FDA.
In December 2013, the agency set out to reduce that number with new guidance requesting pharmaceutical makers begin a three-year phase-out of antibiotics for the specific purpose of helping animals grow faster on less feed, a practice known as “growth promotion.”
The new approach won’t mean an end to antibiotics on the farm, however, as veterinarians and public health officials agree that the drugs are important for treating sick animals. To address the primary drivers of sales — disease prevention and treatment — the FDA is seeking to change the drugs’ over-the-counter status so that farmers will now need a veterinarian’s prescription in order to administer them.
In June, the FDA announced that 31 of the 283 drug products affected by the guidelines have already been withdrawn from the market. But despite this early success, critics have knocked the guidelines for that they consider to be two glaring loopholes.
The first complaint is that the policy is entirely voluntary, meaning drug makers are not legally obligated to comply. Even if they do, critics fear, they could still back out. FDA officials have responded by noting that all 26 companies currently selling growth-promoting antibiotics have promised in writing to change their labels by the end of the phase-in period. And they say, once the labels are changed, veterinarians could lose their license if they break the guidelines.
A second concern is that the recommendations do little to prevent antibiotics that were once sold for growth promotion from being reclassified as drugs that can be used for treatment and prevention.
It’s also unclear how much the guidelines will reduce the overall use of antibiotics on the farm. The FDA doesn’t collect data on how the drugs are being used, and attempts by legislators in Congress to require more specific data reporting have gone nowhere. The U.S. Farmers and Ranchers Alliance, an industry group, estimates that only 13 percent of antibiotic use in food animals is for growth promotion.
Amid such complaints, some lawmakers have pressed the agency for more details. In a July 28, 2014 letter to FDA Commissioner Margaret Hamburg, Sen. Elizabeth Warren (D-Mass.), Sen. Dianne Feinstein (D-Calif.) and Sen. Kirsten Gillibrand (D-NY) expressed concerns that the voluntary guidelines “do not go far enough.” The benefits, they wrote, “will be negligible … if the same animals can continue receiving the same antibiotics at the same doses.”
Part of the FDA’s desire for a voluntary approach stems from challenges the agency has faced in past attempts to restrict antibiotic use on farms. In 1977, the agency tried — and failed — to ban two classes of antibiotics used in animal feeds. In 2000, it tried to withdraw a powerful class of antibiotics, known as fluoroquinolones, from use in poultry, sparking a five-year court battle with Bayer, one of the drug manufacturers. While the FDA prevailed, the effort soured regulators on taking a drug-by-drug approach.
“We actually believe that by taking a voluntary approach we are going to move toward our goal of getting these antibiotics out of use for growth promotion in a more effective and speedier way than if we actually tried to go drug by drug with a formal regulatory process,” FDA Commissioner Hamburg told FRONTLINE. “We always can take further actions as necessary,” she said.
Critics say the voluntary nature of the policy all but ensures more action will be needed. They point to the example of The Netherlands, where usage of antibiotics stayed the same after growth-promoting antibiotics were banned, only to drop 56 percent following the introduction of a ban on the drugs for disease prevention.
But just because a policy is voluntary, doesn’t mean it can’t work. Jacob Gersen, a professor of law at Harvard University and the director of the school’s Food Law Lab, points to the example of the Emergency Planning and Community Right-to-Know Act. The 1986 law asked companies to publicly disclose the release of toxic chemicals from their facilities. Although it did not ban their use in any way, the bad publicity that followed such disclosures is credited with driving down toxic emissions.
One advantage for the FDA, according to Gerson, is that many of the same drug makers that produce antibiotics for food animals are likely to require the agency’s approval for other medications. “The drug companies know that the FDA can make their life really miserable down the road,” he said, which is why they may be more likely to comply.
“Either it’s genius because they have so much sway over the industry that they will get the exact outcome they want … or it’s really, really weak and nothing will happen as a result,” said Gerson. “Time will tell.”

Related Documentaries
Latest Documentaries
Related Stories
Related Stories
Explore
Policies
Teacher Center
Funding for FRONTLINE is provided through the support of PBS viewers and by the Corporation for Public Broadcasting, with major support from Ford Foundation, and The Fialkow Family Foundation. Additional funding is provided the Abrams Foundation, Park Foundation, John D. and Catherine T. MacArthur Foundation, Heising-Simons Foundation, and the FRONTLINE Trust, with major support from Jon and Jo Ann Hagler on behalf of the Jon L. Hagler Foundation, and Corey David Sauer, and additional support from Koo and Patricia Yuen. FRONTLINE is a registered trademark of WGBH Educational Foundation. Web Site Copyright ©1995-2025 WGBH Educational Foundation. PBS is a 501(c)(3) not-for-profit organization.