“Opioids, Inc.”: 10 Key Takeaways From FRONTLINE and the Financial Times’ Insys Investigation
June 23, 2020
Share
What role have pharmaceutical companies played in fueling America’s epidemic of opioid addiction? How have they and their stockholders profited? And how are they being held accountable?
The new FRONTLINE documentary Opioids, Inc., part of a yearlong reporting collaboration with the Financial Times, explores those questions through the story of one company: Insys Therapeutics.
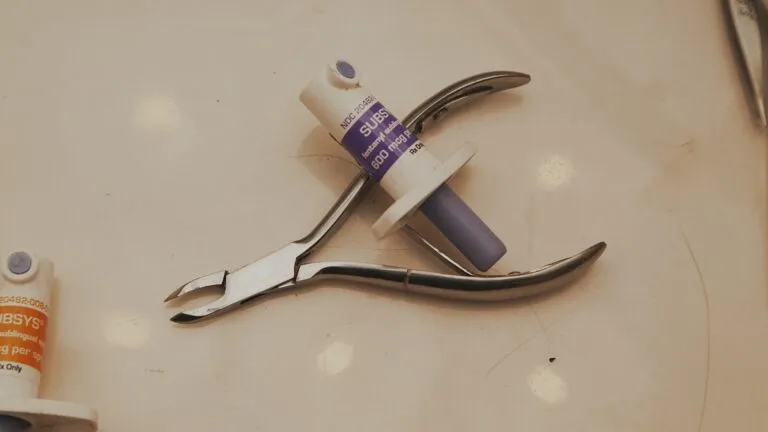
The documentary reveals how Insys bribed doctors, committed insurance fraud and made millions for Wall Street investors pushing a highly addictive opioid painkiller called Subsys — and then, how it became the first pharmaceutical company to have its CEO sentenced to prison time in connection with the opioid crisis.
The documentary is now streaming at the top of this story, on the PBS Video App, on YouTube and on-demand. It airs on PBS stations Tues., June 23 at 10/9c (check local listings). Here are ten key takeaways from FRONTLINE and the Financial Times’ investigation of Insys’ spectacular rise and fall:
1. In the beginning, hopes were high that Subsys, a fast-acting fentanyl-based painkiller, would revolutionize cancer pain treatment—a mission that was personal for Dr. John Kapoor, Insys’ founder.
“I believed that he was somebody that was able to help us change the way we have cancer pain treatment in America,” pain management specialist Dr. Lisa Jo Stearns, who ran a clinical trial of Subsys, says in the film of her initial impression of Kapoor. She recalls him tearing up as he discussed the pain experienced by his wife as she died of cancer.
2. But after a slow initial launch, insiders say the business model became focused on generating prescriptions and sales “by any means necessary.”
“It wasn’t about cancer patients. It was about getting as many people as you could on the drug,” says former sales representative April Moore.
3. Alec Burlakoff, Insys’ former head of sales, admits on camera to bribing doctors — in order, he says, to meet Kapoor’s profit imperative.
“The instruction was, go out there, show that we can get a minimum return on investment of 2:1, minimum, and do not lose [Kapoor’s] money or get fired. And the only way that I knew how to do it to get that guarantee, is to bribe doctors,” Burlakoff tells FRONTLINE and the FT in his first on-camera interview.
4. Burlakoff’s team targeted high-prescribing doctors and nurse practitioners known within the company as “whales,” and pressured them to prescribe ever-higher doses of Subsys in a process known as “titration.”
The company even held contests for the sales team: the higher the prescription doses they got doctors to write, the larger the cash prize — despite the dangers to patients. “This is probably the most guilty thing in the company. If you had a doctor who wrote a prescription, it was a bonus on your paycheck. If you reach the 1600 mcg of fentanyl, that could kill you, but people were getting there,” says former sales representative, Tim Neely.
5. Insys hired inexperienced people for sales and senior management positions.
“A saleswoman told me about Sunrise Lee, who oversaw the entire Midwest. The only Sunrise Lee that I could track had been a dancer at a gentleman’s club in Florida. She has no pharmaceutical background, no science background,” journalist Roddy Boyd says. Lee wouldn’t speak to FRONTLINE, but when Boyd was reporting, she said that she was under federal investigation for Insys sales practices. “I was not nearly as concerned at hiring a former Playboy model or a former exotic dancer as I was assessing whether or not they had what I call, unfortunately, ‘a killer instinct’ — almost no conscience,” Burlakoff says.
6. The company misled insurers to cover prescriptions of Subsys, which was only approved by the FDA for treating breakthrough cancer pain.
“None of what we were saying was truthful,” a former prior authorization specialist says. “We’re just pocketing the money off of a prescription that should’ve never been approved anyway. That’s insurance fraud.”
7. Many Wall Street investors continued to propel Insys’ success, even as questions about its business practices mounted.
“There were people on Wall Street who were intent on keeping this stock price up, and I believe ignoring some of the more obvious indications that this was an unsustainable, potentially fraudulent business model,” says Jim Carruthers, one of a small number of investors known as short sellers who were skeptical of Insys.
8. Federal prosecutors brought Insys down in court using laws originally designed to catch mob bosses.
Using anti-racketeering laws, prosecutors worked their way up the company’s ranks. They ultimately arrived at a “smoking gun”: a spreadsheet ordered by Kapoor that showed how Insys tracked the money that went to doctors, and what the company got in return. “I called it a smoking gun. I mean, it was about this return on investment theory that Kapoor had, and that’s criminal. So you might as well be writing out a confession,” says federal prosecutor Fred Wyshak, who had previously been renowned for prosecuting mob kingpins like Whitey Bulger.
9. Other opioid makers are now on notice.
With prison terms handed down for Kapoor, Burlakoff and other former Insys executives, other drug makers and distributors are in the hot seat as the opioid crisis continues. “When executives go to jail, that sends out a pretty serious message to the wider business and financial community,” says Gillian Tett, FT editor-at-large, US. “And certainly with the Insys story, that message will be pretty clear-cut to the rest of the pharmaceutical world.”
10. Subsys has been linked to hundreds of deaths.
One grieving mother hopes the prison sentences for Insys executives will make other drugmakers think twice about pushing opioids: “Normally, they just get a slap on the wrist and have to pay a penalty, which was easily made up by selling more opioids. At least now, they’ll have to think, maybe they’ll go to jail for this,” Deborah Fuller, whose 32-year-old daughter Sarah died from an overdose of drugs that included Subsys, told the Financial Times in a written story published in tandem with the documentary.
For the full story, watch the Opioids, Inc. documentary, read the FT’s in-depth text piece, and listen to a conversation between Opioids, Inc filmmaker Tom Jennings and FT reporter Hannah Kuchler on the FRONTLINE Dispatch.
This story has been updated to correct Gillian Tett’s title at the FT from editor to editor-at-large, US.

Related Documentaries
Latest Documentaries
Related Stories
Related Stories
Explore
Policies
Teacher Center
Funding for FRONTLINE is provided through the support of PBS viewers and by the Corporation for Public Broadcasting, with major support from Ford Foundation, and The Fialkow Family Foundation. Additional funding is provided the Abrams Foundation, Park Foundation, John D. and Catherine T. MacArthur Foundation, Heising-Simons Foundation, and the FRONTLINE Trust, with major support from Jon and Jo Ann Hagler on behalf of the Jon L. Hagler Foundation, and Corey David Sauer, and additional support from Koo and Patricia Yuen. FRONTLINE is a registered trademark of WGBH Educational Foundation. Web Site Copyright ©1995-2025 WGBH Educational Foundation. PBS is a 501(c)(3) not-for-profit organization.