White House Eases Cap on Medication for Opioid Addiction
July 6, 2016
Share
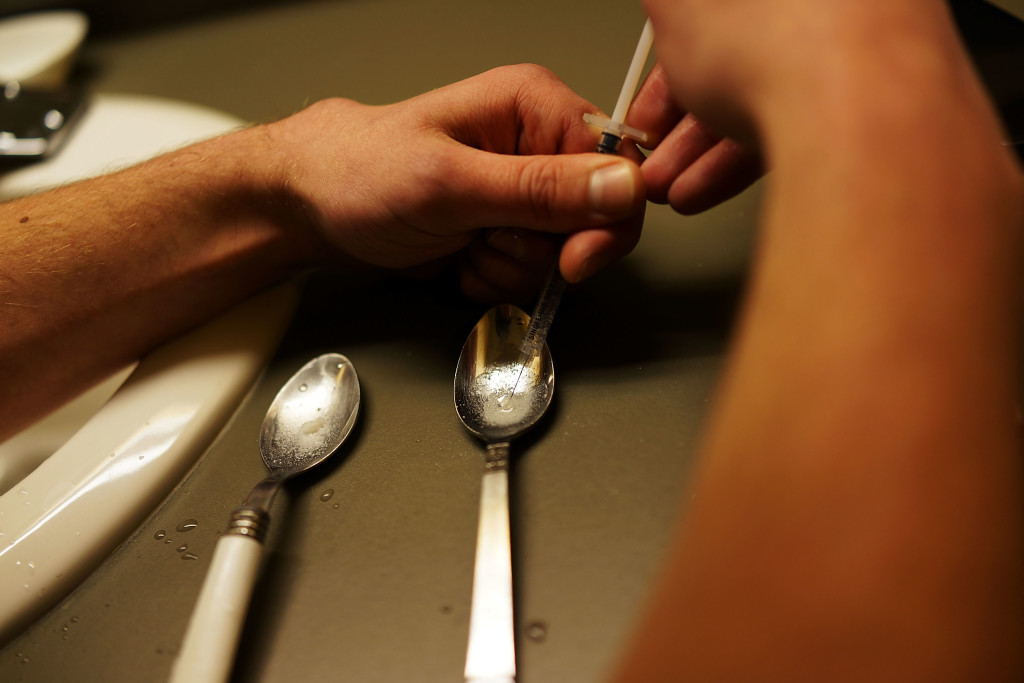
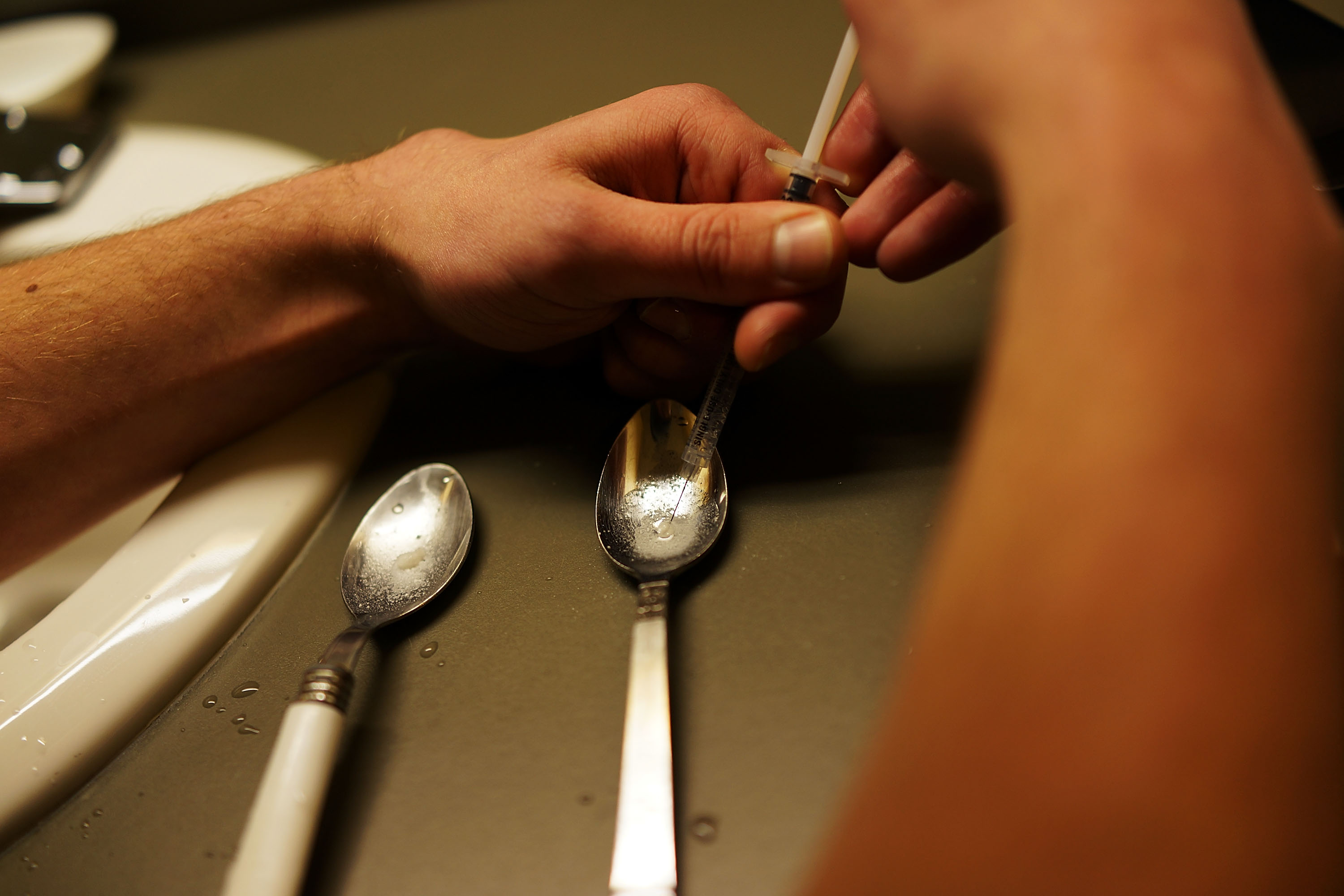
Locked in a fight with Congress over $1.1 billion in new funding to address prescription opioid and heroin abuse, the Obama administration announced Wednesday that it will ease federal controls on a medication prescribed by doctors to treat addiction.
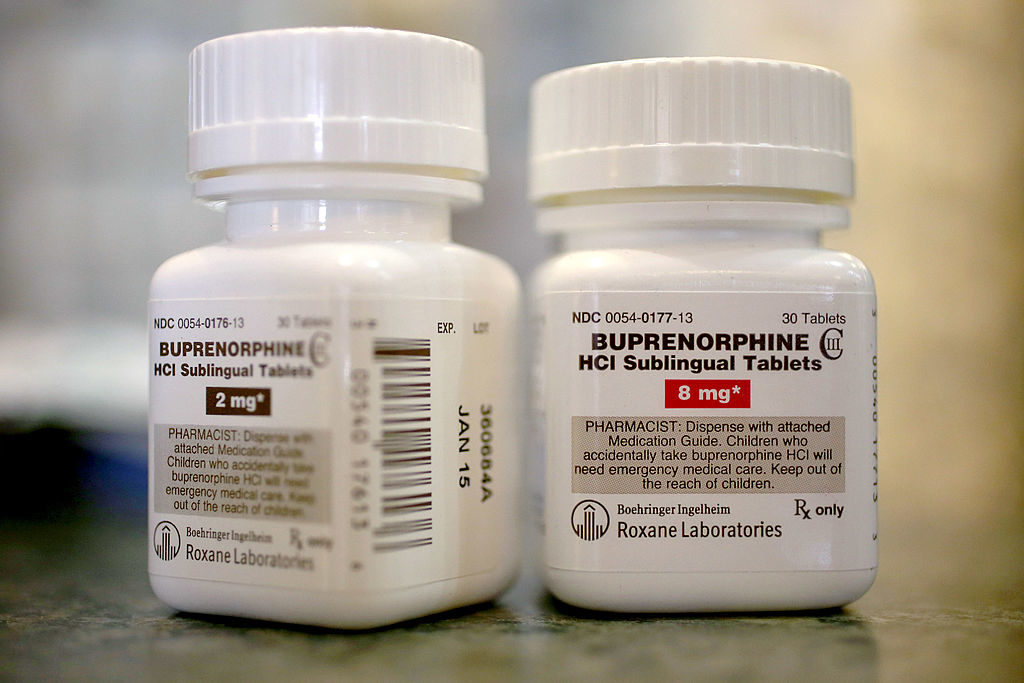
The Department of Health and Human Services will now allow doctors to prescribe buprenorphine, a drug approved to treat opioid addiction, to 275 patients at once. Previously, providers could prescribe to no more than 100 patients at a time.
Because the medication is itself a narcotic, federal authorities have said the cap was meant to prevent buprenorphine’s proliferation in the black market and abuse by people suffering from addiction.
But at a time when more than 47,000 Americans a year are dying from drug overdoses — more than are killed in automobile accidents – critics say the limits have made it needlessly more difficult for patients to access one of the more successful forms of treatment available. Buprenorphine is considered a safer and more effective treatment option for patients with mild to moderate dependence, and is often held up as a treatment option for people who live too far from methadone clinics.
In order to prescribe buprenorphine, physicians must first receive a waiver from the Drug Enforcement Agency, after which they can only write 30 prescriptions the first year. Even if a doctor has the waiver, many are hesitant to prescribe the medication, creating long waiting lists.
Roughly 33,600 physicians currently have waivers to prescribe the drug, according to federal data — with 67.5 percent limited to treat 30 patients at once, and the remaining 32.5 percent allowed to treat 100.
Approximately 650,000 Americans were prescribed buprenorphine in 2014. When the change takes effect in August, as many as 90,000 more could gain access, officials from the Department of Health and Human Services and the White House Office of National Drug Control Policy told reporters on a conference call.
Dr. Jeffrey Goldsmith, president of the American Society of Addiction Medicine, lauded the shift in policy.
“For too long, addiction specialists like me have had to turn patients in need away from treatment that might save their lives, not because we don’t have the expertise or capacity to treat them, but because of an arbitrary federal limit,” Goldsmith said in a statement. “It has been heartbreaking to tell patients and their families that my hands are tied, and to see them continue to suffer from a treatable disease.”
The administration also announced new rules aimed at improving drug monitoring by federal prescribers treating Native Americans and military veterans; a proposal to end financial incentives based on patient feedback for doctors who prescribe opioids; and more than a dozen new scientific studies on pain treatment and opioid dependence.
The changes come as lawmakers in Congress are meeting to finalize legislation aimed at tackling the nation’s deadly opioid epidemic. The White House is requesting $1.1 billion to address the crisis, while Democrats on Capitol Hill are calling for $920 million in funding. The GOP has offered $581 million in new spending to address opioid abuse, but on Wednesday White House Press Secretary Josh Earnest said, “If there is a bill that reaches the president’s desk that is geared toward fighting the opioid epidemic but doesn’t include additional funding, I certainly cannot promise that the president would sign it.”
Related Documentaries
Latest Documentaries
Related Stories
Related Stories
Explore
Policies
Teacher Center
Funding for FRONTLINE is provided through the support of PBS viewers and by the Corporation for Public Broadcasting, with major support from Ford Foundation, and The Fialkow Family Foundation, as part of the Plum Bush Foundation. Additional funding is provided the Abrams Foundation, Park Foundation, John D. and Catherine T. MacArthur Foundation, Heising-Simons Foundation, and the FRONTLINE Trust, with major support from Jon and Jo Ann Hagler on behalf of the Jon L. Hagler Foundation, and Corey David Sauer, and additional support from Koo and Patricia Yuen. FRONTLINE is a registered trademark of WGBH Educational Foundation. Web Site Copyright ©1995-2025 WGBH Educational Foundation. PBS is a 501(c)(3) not-for-profit organization.